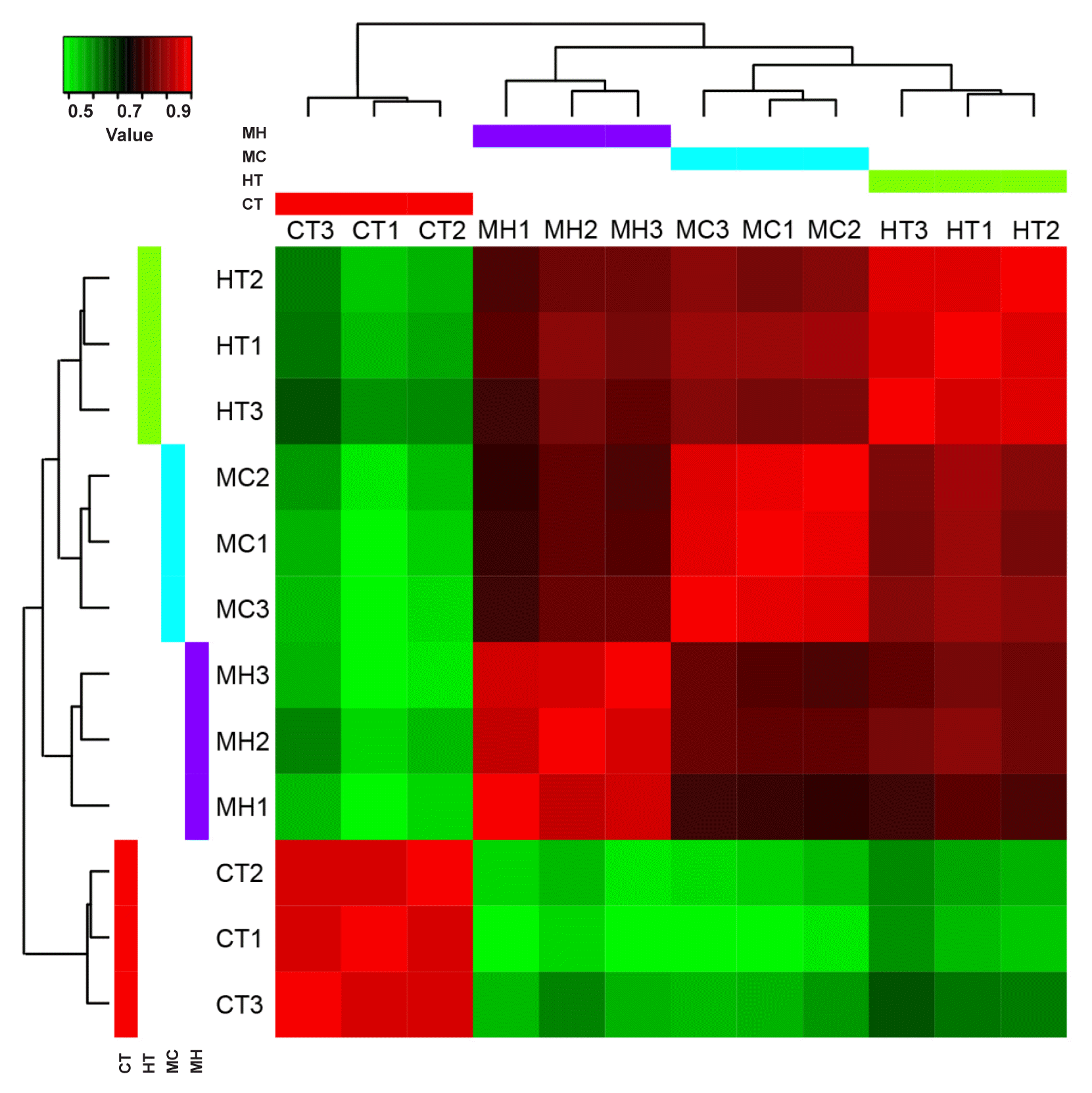
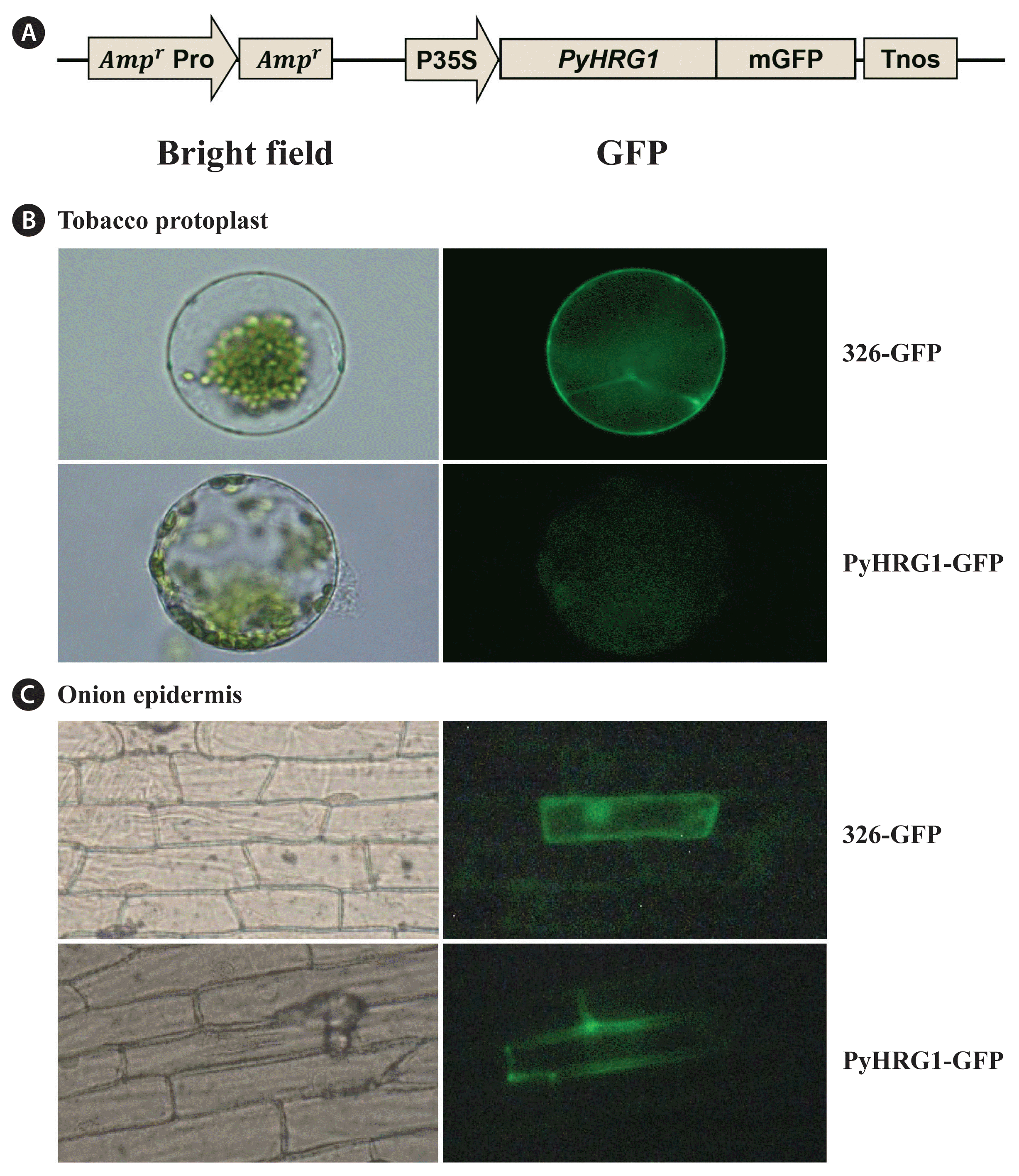
Avila, M, Santelices, B & McLachlan, J 1986. Photoperiod and temperature regulation of the life history of
Porphyra columbina (Rhodophyta, Bangilaes) from central Chile. Can J Bot. 64:1867–1872.

Blouin, NA, Brodie, JA, Grossman, AC, Xu, P & Brawley, SH 2011.
Porphyra: a marine crop shaped by stress. Trends Plant Sci. 16:29–37.

Chan, CX, Blouin, NA, Zhuang, Y, Zäuner, S, Prochnik, SE, Lindquist, E, Lin, S, Benning, C, Lohr, M, Yarish, C, Gantt, E, Grossman, AR, Lu, S, Müller, K, Stiller, JW, Brawley, SH & Bhattacharya, D 2012.
Porphyra (Bangiophyceae) transcriptomes provide insights into red algal development and metabolism. J Phycol. 48:1328–1342.


Chen, S & Li, H 2017. Heat stress regulates the expression of genes at transcriptional and post-transcriptional levels, revealed by RNA-seq in
Brachypodium distachyon
. Front Plant Sci. 7:2067 pp.


Choi, JY, Seo, YS, Kim, SJ, Kim, WT & Shin, JS 2011. Constitutive expression of
CaXTH 3, a hot pepper xyloglucan endotransglucosylase/hydrolase, enhanced tolerance to salt and drought stresses without phenotypic defects in tomato plants (
Solanum lycopersicum cv. Dotaerang). Plant Cell Rep. 30:867–877.


Choi, S, Hwang, MS, Im, S, Kim, N, Jeong, W-J, Park, E-J, Gong, Y-G & Choi, D-W 2013. Transcriptome sequencing and comparative analysis of the gametophyte of
Pyropia tenera under normal and high-temperature condition. J Appl Phycol. 25:1237–1246.

Dai, YL, Kim, GH, Kang, MC & Jeon, YJ 2020. Protective effects of extracts from six local strains of
Pyropia yezoensis against oxidative damage
in vitro and in zebrafish model. Algae. 35:189–200.

Ha, YI, Lim, JM, Ko, SM, Liu, JR & Choi, D-W 2007. A ginseng-specific abundant protein (GSAP) located on the cell wall is involved in abiotic stress tolerance. Gene. 386:115–122.


Hwang, EK & Park, CS 2020. Seaweed cultivation and utilization of Korea. Algae. 35:107–121.

Hwang, MS, Chung, I-K & Oh, Y-S 1997. Temperature responses of Porphyra tenera Kjellman and P. yezoensis Ueda (Bangiales, Rhodophyta) from Korea. Algae. 12:207–213.
Im, S, Choi, S, Hwang, MS, Park, E-J, Jeong, W-J & Choi, D-W 2015.
De novo assembly of transcriptome from the gametophyte of the marine red algae
Pyropia seriata and identification of abiotic stress response genes. J Appl Phycol. 27:1343–1353.

Im, S, Lee, H-N, Jung, HS, Yang, S, Park, E-J, Hwang, MS, Jeong, W-J & Choi, D-W 2017. Transcriptome-based identification of the desiccation response genes in marine red algae
Pyropia tenera (Rhodophyta) and enhancement of abiotic stress tolerance by
PtDRG2 in
Chlamydomonas
. Mar Biotechnol. 19:232–245.

Kim, M, Wi, J, Lee, J, Cho, W-B, Park, E-J, Hwang, M-S, Choi, S-J, Jeong, W-J, Kim, GH & Choi, D-W 2021. Development of genomic simple sequence repeat (SSR) markers of
Pyropia yezoensis (Bangiales, Rhodophyta) and evaluation of genetic diversity of Korean cultivars. J Appl Phycol, Advanced online publication,
https://doi.org/10.1007/s10811-021-05236-7

Le Gall, H, Philippe, F, Domon, J-M, Gillet, F, Pelloux, J & Rayon, C 2015. Call wall metabolism in response to abiotic stress. Plants. 4:112–166.


Lu, Y & Xu, J 2015. Phytohormones in microalgae: a new opportunity for microalgal biotechnology? Trends Plant Sci. 20:273–282.


Luo, Q, Zhu, Z, Zhu, Z, Yang, R, Qian, F, Chen, H & Yan, X 2014. Different responses to heat shock stress revealed heteromorphic adaptation strategy of
Pyropia haitanensis (Bangiales, Rhodophyta). PLoS ONE. 9:e94354 pp.



McLachlan, J 1973. Growth media-marine. In : Stein JR, editor Handbook of Phycological Methods: Culture Methods and Growth Measurements. Cambridge University Press, New York, 25–51.
Mittler, R 2002. Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci. 7:405–410.


Mittler, R, Vanderauwera, S, Gollery, M & Van Breusegem, F 2004. Reactive oxygen gene network of plants. Trends Plant Sci. 9:490–498.


Moore, JP, Vicré-Gibouin, M, Farrant, JM & Driouich, A 2008. Adaptations of higher plant cell walls to water loss: drought vs. desiccation. Physiol Plant. 134:237–245.


Na, Y, Lee, H-N, Wi, J, Jeong, W-J & Choi, D-W 2018.
PtDRG1, a desiccation response gene from
Pyropia tenera (Rhodophyta), exhibits chaperone function and enhances abiotic stress tolerance. Mar Biotechnol. 20:584–593.

Rienth, M, Torregrosa, L, Luchaire, N, Chatbanyong, R, Lecourieux, D, Kelly, MT & Romieu, C 2013. Day and night heat stress trigger different transcriptomic responses in green and ripening grapevine (Vitis vinifera) fruit. BMC Plant Biol. 14:108 pp.
Rodriguez, MCS, Edsgärd, D, Hussain, SS, Alquezar, D, Rasmussen, M, Gilbert, T, Nielsen, BH, Bartels, D & Mundy, J 2010. Transcriptomes of the desiccation-tolerant resurrection plant
Craterostigma plantagineum
. Plant J. 63:212–228.


Sasidharan, R, Voesenek, LACJ & Pierik, R 2011. Cell wall modifying proteins mediate plant acclimatization to biotic and abiotic stresses. Crit Rev Plant Sci. 30:548–562.

Shin, YJ, Min, SR, Kang, DY, Lim, J-M, Park, E-J, Hwang, MS, Choi, D-W, Ahn, J-W, Park, Y-I & Jeong, W-J 2018. Characterization of high temperature-tolerant strains of
Pyropia yezoensis
. Plant Biotechnol Rep. 12:365–373.

Shinozaki, K, Uemura, M, Bailey-Serres, J, Bray, EA, Bailey-Serres, J & Weretilnyk, E 2015. Responses to abiotic stresses. In : Buchanan B, Gruissem W, Jones R, editors Biochemistry and Molecular Biology of Plants. American Society of Plant Biologist, Rockville, MD, 1051–1100.
Song, J, Liu, Q, Hu, B & Wu, W 2016. Comparative transcriptome profiling of
Arabidopsis Col-0 in responses to heat stress under different light conditions. Plant Growth Regul. 79:209–218.

Van den Ende, W & Valluru, R 2009. Sucrose, sucrosyl oligosaccharides, and oxidative stress: scavenging and salvaging? J Exp Bot. 60:9–18.


Wahid, A, Gelani, S, Ashraf, M & Foolad, MR 2007. Heat tolerance in plants: an overview. Environ Exp Bot. 61:199–233.

Wang, K, Liu, Y, Tian, J, Huang, K, Shi, T, Dai, X & Zhang, W 2017. Transcriptional profiling and identification of heat-responsive genes in perennial ryegrass by RNA-sequencing. Front Plant Sci. 8:1032 pp.



Wang, W, Vinocur, B, Shoseyov, O & Altman, A 2004. Role of plant heat-shock proteins and molecular chaperons in the abiotic stress response. Trends Plant Sci. 9:244–252.


Yang, KA, Lim, CJ, Hong, JK, Park, CY, Cheong, YH, Chung, WS, Lee, KO, Lee, SY, Cho, MJ & Lim, CO 2006. Identification of cell wall genes modified by a permissive high temperature in Chinese cabbage. Plant Sci. 171:175–182.