Merging the cryptic genera Radicilingua and Calonitophyllum (Delesseriaceae, Rhodophyta): molecular phylogeny and taxonomic revision
Article information
Abstract
Radicilingua Papenfuss and Calonitophyllum Aregood are two small genera of the family Delesseriaceae that consist of only three and one taxonomically accepted species, respectively. The type species of these genera, Radicilingua thysanorhizans from England and Calonitophyllum medium from the Americas, are morphologically very similar, with the only recognized differences being vein size and procarp development. To date, only other two species were recognized inside the genus Radicilingua: R. adriatica and R. reptans. In this study, we analysed specimens of Radicilingua collected in the Adriatic and Ionian Sea (Mediterranean), including a syntype locality of R. adriatica (Trieste, northern Adriatic Sea), alongside material from near the type locality of R. thysanorhizans (Torpoint, Cornwall, UK). The sequences of the rbcL-5P gene fragment here produced represent the first molecular data available for the genus Radicilingua. Phylogenetic reconstruction showed that the specimens from the Adriatic and Ionian Seas were genetically distinct from the Atlantic R. thysanorhizans, even if morphologically overlapping with this species. A detailed morphological description of the Mediterranean specimens, together with an accurate literature search, suggested that they were distinct also from R. adriatica and R. reptans. For these reasons, a new species was here described to encompass the Mediterranean specimens investigated in this study: R. mediterranea Wolf, Sciuto & Sfriso. Moreover, in the rbcL-5P tree, sequences of the genera Radicilingua and Calonitophyllum grouped in a well-supported clade, distinct from the other genera of the subfamily Nitophylloideae, leading us to propose that Calonitophyllum medium should be transferred to Radicilingua.
INTRODUCTION
Radicilingua Papenfuss (1956) is a small genus in the family Delesseriaceae with three currently accepted species: the type species Radicilingua thysanorhizans (Holmes) Papenfuss, R. adriatica (Kylin) Papenfuss and R. reptans (Kylin) Papenfuss (Guiry and Guiry 2020). Radicilingua has been reported along the Mediterranean and northeastern Atlantic coasts (Guiry and Guiry 2020). Calonitophyllum Aregood (1975) is a monotypic genus in the same family, based on Calonitophyllum medium (Hoyt) Aregood from North and South America, which has never been found in the Mediterranean Sea.
Detailed morphological analyses were previously conducted on the type species of Radicilingua. Kylin (1924) carefully analysed the growth of R. thysanorhizans (as Rhizoglossum thysanorhizans [Holmes] Kylin) and described the differentiation of the characteristic microscopic blade veins; Huvè and Riouall (1970) illustrated young plants, marginal proliferations and rhizoid formation. Kylin (1956) observed the reproductive structures and reported scattered cystocarps, gonimoblasts bearing abundant carpospores and tetrasporangia in sori near the thallus tips. A detailed description of English specimens was also reported by Maggs and Hommersand (1993). Conversely, the other two species of Radicilingua, R. adriatica and R. reptans, reported from the Mediterranean Sea in several checklists (e.g., Gómez Garreta et al. 2001, Sfriso and Curiel 2007, Bottalico et al. 2016, Gallardo et al. 2016, Petrocelli et al. 2019), are inadequately known. Another hindrance to correct identification is the absence of DNA sequences for Radicilingua in public databases.
Morphological and reproductive characters of C. medium were reported by Aregood (1975). C. medium resembles R. thysanorhizans in its vegetative development, but it is distinguishable by very prominent macroscopic veins and by its reproductive structures. Aregood (1975) reported that the procarp of C. medium is characterized by a single carpogonial filament and a single large persistent sterile cell, which is absent in R. thysanorhizans. This character is not easy to observe, especially in thalli that are not fully mature. Moreover, the formation of macroscopic veins is restricted to extreme basal regions and does not occur in males or young thalli (Aregood 1975). For these reasons, the distinction between species belonging to these genera is very difficult.
The goals of this study were to produce the first molecular data available for the genus Radicilingua and to compare Atlantic and Mediterranean specimens. Moreover, the phylogenetic relationship between the morphologically overlapping genera Radicilingua and Calonitophyllum was investigated.
MATERIALS AND METHODS
Sampling
Specimens morphologically identified as Radicilingua sp. were collected from December 2019 to April 2020 in the Adriatic and Ionian Seas: three sampling sites in the Venice lagoon (station 1: 45°25′35.2″ N, 12°24′49.0″ E; station 2: 45°19′38.9″ N, 12°19′46.1″ E; station 3: 45°16′16.5″ N, 12°15′0.32″ E), one in Trieste (station 4: 45°38′10.5″ N, 13°45′27.6″ E) and one in the Mar Piccolo of Taranto (station 5: 40°29′39″ N, 17°19′22″ E). In March 2021, cystocarpic plants were also found in the Mar Piccolo of Taranto (station 5).
Two specimens of the type species of Radicilingua, R. thysanorhizans, were supplied from the personal herbarium of phycologist Annette (Netty) Elizabeth Little, née Twitchin (born Nov 4, 1948, died Jul 15, 2002), which her husband David Little donated, after her death, to the British Phycological Society. The two analyzed specimens were collected in July 1986 from Duke Rock, Plymouth, England, near the type locality of R. thysanorhizans, and in August 1984 in Pembrokeshire, Wales (Fig. 1A & B), respectively.
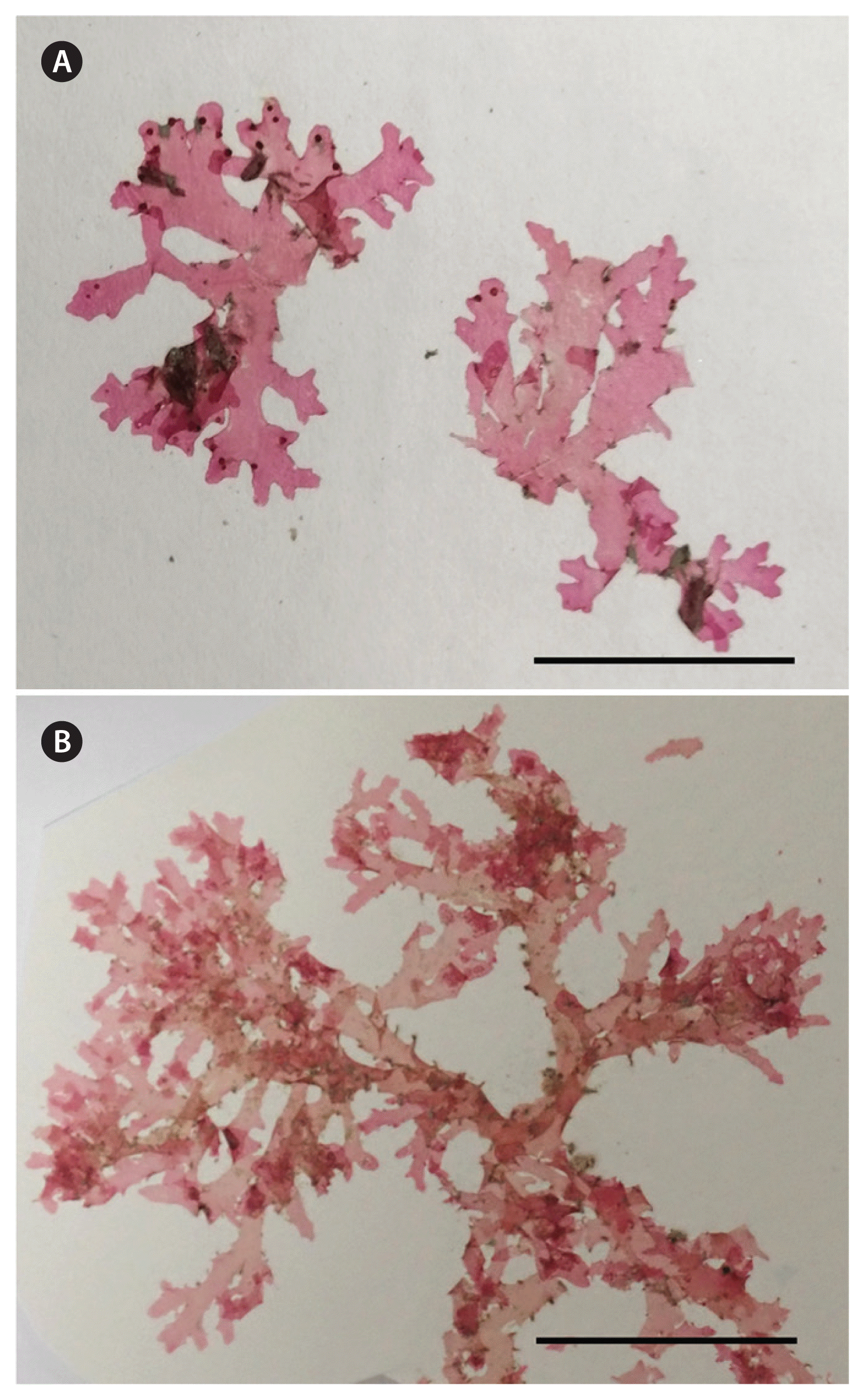
Radicilingua thysanorhizans (Holmes) Papenfuss. (A) Habit of specimens collected in Pembrokeshire (United Kingdom) in August 1984, leg: A. E. Little, det: C. A. Maggs. (B) Habit of the specimen collected in Plymouth (United Kingdom) in July 1986, leg: A. E. Little, det: C. A. Maggs. Scale bars represent: A & B, 25 mm.
Molecular analyses
Genomic DNA was extracted using the Genomic DNA purification kit (Thermo Fisher Scientific, Waltham, MA, USA). Amplification of the rbcL-5P gene fragment (about 700 bp) was carried out with the primer pairs F57-R753 (Freshwater and Rueness 1994), following Wolf et al. (2018). The obtained PCR products were cleaned using the HT ExoSAP-IT (Thermo Fisher Scientific) and sequencing was carried out at the Eurofins Genomics Sequencing Service (Eurofins Genomics GmbH, Ebersberg, Germany), with the same primers employed in the amplification reactions. The GeneStudio sequence analysis software (http://genestudio.com/) was used to assemble the final consensus sequences. The new sequences (one for each sampling site) were deposited in the International Nucleotide Sequence Database Collaboration (INSDC) repositories, through the European Nucleotide Archive (ENA) platform, with the following accession numbers: LR991380-LR991386.
Using the BLAST program, available at the USA National Center for Biotechnology Information (NCBI) web server (http://www.ncbi.nlm.nih.gov), the obtained sequences were compared with those present in the INSDC archives.
A dataset of rbcL-5P sequences was created including the newly obtained sequences and other suitable sequences available in the INSDC repositories, following the most recent classifications for the focal taxa (Lin et al. 2001, 2004a, 2004b, Lin and Fredericq 2003, Díaz-Tapia et al. 2019). Representative sequences of the family Delesseriaceae were included, and in particular sequences of the subfamilies Nitophylloideae, Sarcomenioideae, Delesserioideae, and Phycodryoideae. Two representative sequences of the Rhodomelaceae (Odonthalia dentata, [Linnaeus] Lyngbye, INSDC accession: KU564463, and Rhodomela confervoides [Hudson] P. C. Silva, INSDC accession: MH277301) were used as the outgroup to orient the tree. Supplementary Table S1 lists the Delesseriaceae specimens with rbcL-5P sequences included in the phylogenetic analyses.
A multiple sequence alignment was generated with MUSCLE (Edgar 2004) and used for phylogenetic analyses; it included 58 sequences for a total of 630 aligned positions. Phylogenetic analyses based on the neighbour joining (NJ), maximum parsimony (MP), and maximum likelihood (ML) methods were performed with MEGA X (Kumar et al. 2018). For ML, the model that best fit the data was GTR + G + I, as suggested by the “Find best DNA Models” tool implemented in MEGA under the Bayesian information criterion (Schwarz 1978). A non-parametric bootstrap re-sampling (Felsenstein 1985) of 1,000 replicates was performed to test the robustness of the tree topologies. Bayesian inference (BI) analyses were carried out with MrBayes version 3.1.2 (Ronquist and Huelsenbeck 2003). The analyses included two independent Markov chain Monte Carlo (MCMC) runs, each composed of four chains (three heated and one cold); each MCMC ran for 2 × 106 generations, sampling trees every 100 generations. The sampling of the posterior distribution was considered to be adequate if the average standard deviation of split frequencies was ≤0.01. The first 5,000 trees were discarded as burn-in, as determined by stationarity of log likelihood assessed using Tracer version 1.5 (Rambaut and Drummond 2007). The consensus topology and posterior probability values were then calculated from the remaining trees. The final tree figure was created with Inkscape v. 0.92. An alignment of the rbcL-5P sequences of only the subfamily Nitophylloideae was also obtained with MUSCLE to calculate the percent identities within this group (Supplementary Table S2).
Morphological analyses
Specimens were preserved in 4% formaldehyde/seawater solution and were observed using a light microscope Optika B-510PH (Optika Srl, Ponteranica, BG, Italy) equipped with a digital image acquisition system. Final pictures, suitable for publication, were created with GIMP v. 2.8.22 (https://www.gimp.org) and Inkscape v. 0.92 (https://www.inkscape.org).
RESULTS
Molecular analyses
The phylogenetic reconstruction based on the rbcL-5P gene fragment (Fig. 2) highlighted different subfamilies and corresponding tribes of the family Delesseriaceae. Of these, the subfamily Nitophylloideae was not monophyletic, except for the topologies obtained with MP and BI methods (data not shown). The sequences of the Mediterranean Radicilingua specimens formed a highly supported clade (100 / 100 / 100 / 1.00), sister to another highly supported group (100 / 100 / 100 / 1.00) consisting of the two R. thysanorhizans specimens from England. The group formed by the Mediterranean and the English Radicilingua clades was sister taxon with strong statistical support (100 / 95 / 100 / 1.00) to a highly supported cluster (99 / 99 / 100 / 1.00) formed by two Calonitophyllum medium sequences from the United States. The two C. medium rbcL sequences were deposited in GenBank by Lin et al. (2001) from specimens collected in North Carolina (type locality of the species) and Texas. The similarities among the rbcL-5P sequences of the specimens belonging to the Radicilingua and Calonitophyllum clades were 96.35–96.67% (i.e., the nucleotide divergence was 3.33–3.65%). The rbcL-5P similarities among the sequences of the remaining Nitophylloideae clades, identified at the genus level and including different species of the corresponding genera, were: 92.86% for Nitophyllum Greville, 95.87–96.51% for Augophyllum S. M. Lin, Fredericq & Hommersand, 96.03% for Valeriemaya A. J. K. Millar & Wynne and 87.78–98.41% for Martensia K. Hering (Supplementary Table S2). Except for the two American specimens of C. medium, which were 99.84% similar (one nucleotide difference in 630 aligned positions), all the other Nitophylloideae specimens attributed to the same species showed 100% rbcL-5P sequence identity.
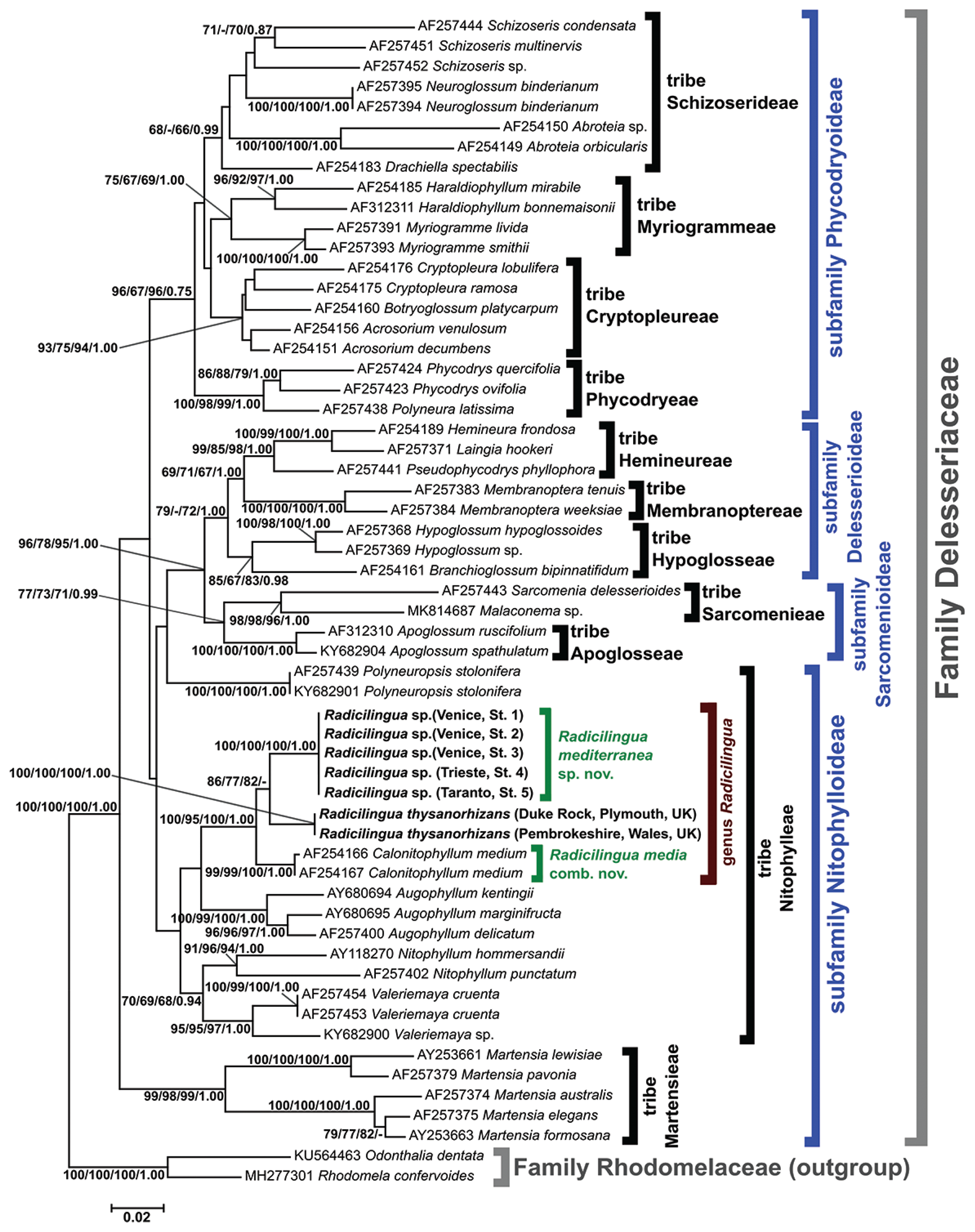
Phylogenetic reconstruction of the family Delesseriaceae based on the partial rbcL gene. The topology obtained with neighbour joining (NJ) method is depicted and the support values from NJ bootstrap, maximum parsimony bootstrap, maximum likelihood bootstrap and Bayesian inference posterior probabilities are reported, respectively. Only bootstrap supports ≥50% and posterior probabilities ≥0.70 are shown. Values for nodes that obtained support in only two of the phylogenetic analyses were omitted. For each of the downloaded sequences, the International Nucleotide Sequence Database Collaboration (INSDC) accession number is reported followed by the species name. The sequences obtained in this work are in boldface font. Square brackets indicate families, subfamilies and tribes. Scale bar represents: expected number of nucleotide substitutions per site.
Based on the obtained molecular and phylogenetic data, the following taxonomic change is proposed.
Radicilingua media (Hoyt) Wolf, Sciuto & Sfriso, comb. nov
Basionym
Nitophyllum medium Hoyt 1920, Bull. U. S. Bur. Fish. 36: 494, pl. CV; pl. CXIV: figs 4–5; text-fig. 35.
Synonyms
Calonitophyllum medium (Hoyt) Aregood, 1975; Hymenena media (Hoyt) W. R. Taylor 1960.
Holotype
United States National Museum, Jul 12, 1907, No. 2, leg: W. D. Hoyt.
Type locality
Bogue Beach, Beaufort, North Carolina.
Distribution
North America (North Carolina, South Carolina, Florida, Texas) and South America (Venezuela).
Description
Aregood (1975): “Plants large (to 20 cm high), rosy-red blades comprised of narrow (0-5-1.5 cm wide) branches; holdfast a prominent branched and somewhat calcified, perennial structure, creamy-pink in colour; tetrasporophytes with conspicuous veins, large and proliferous, spores borne in sori scattered over entire surface of plant; cystocarps also widely scattered, abundant; procarps formed on one or both sides of fertile segment, each comprised of one carpogonial branch and a single very large sterile cell; fertile segment also bears a sterile pericentral cell in addition to the supporting cell; gametophytes somewhat dimorphic; males lack macroscopic veins, are smaller and nearly dichotomous.”
Based on the obtained molecular and phylogenetic data and on the morphological evidence reported below, to encompass the Adriatic and Ionian specimens the following new species is here described.
Radicilingua mediterranea Wolf, Sciuto & Sfriso sp. nov. (Figs 3–5)
Radicilingua mediterranea sp. nov., vegetative morphology. (A) Habit of the thallus. (B) Irregular microscopic veins (arrowheads). (C) Margin of a young blade showing the apical cell (arrowhead). (D) Surface view of parietal discoid plastids (arrowheads). (E) Detail of the margin of a blade showing irregular surface cells (arrowheads). (F) Marginal rhizoidal filaments (arrowheads). (G) Detail of a single rhizoidal filament. (H) Young new blade arising from surface cells of the holdfasts. Scale bars represent: A, 25 mm; B, 200 μm; C–F, 50 μm; G & H, 100 μm.
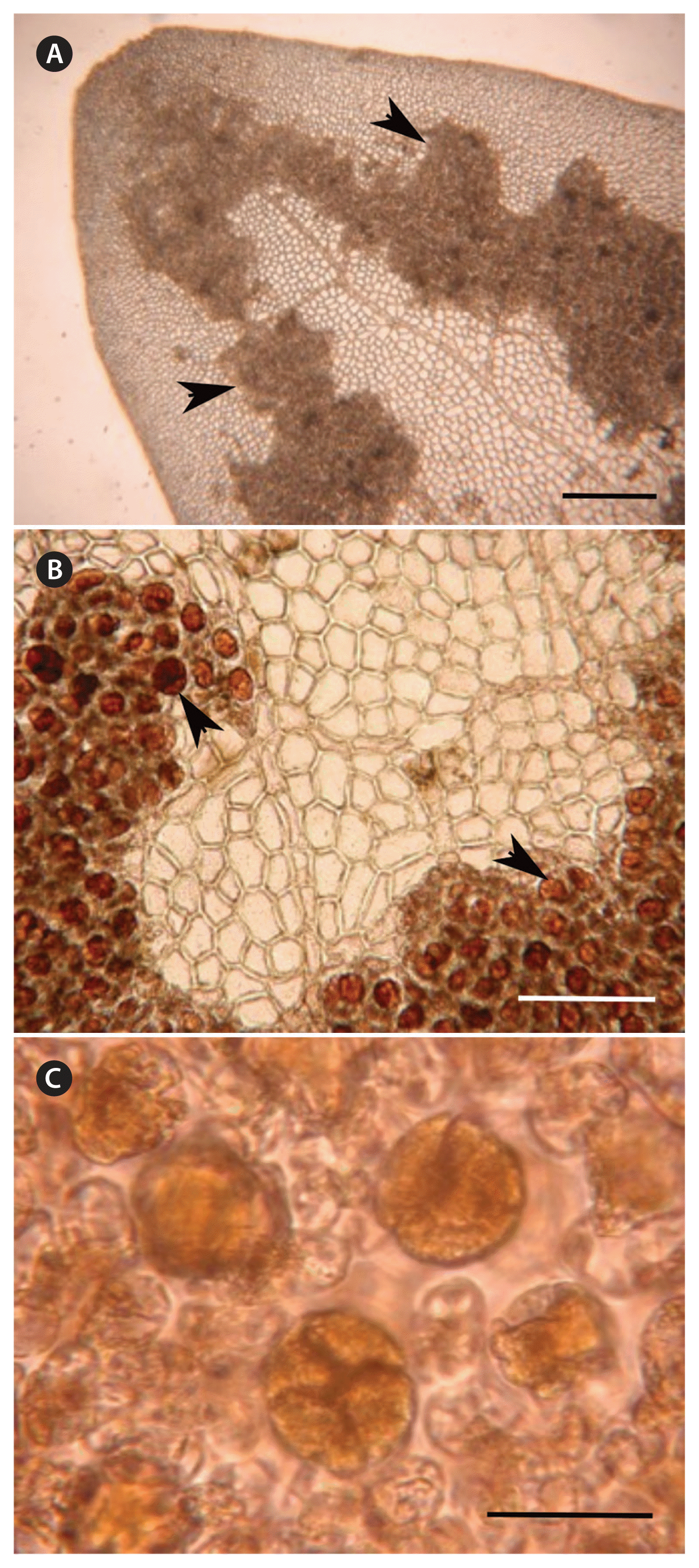
Radicilingua mediterranea sp. nov., tetrasporangial morphology. (A) Surface view of mature tetrasporangial sori scattered over a fertile blade (arrowheads). (B) Detail of the tetrasporangial sori showing numerous tetrasporangia (arrowheads). (C) Detail of mature tetrasporangia. Scale bars represent: A, 100 μm; B, 200 μm; C, 50 μm.
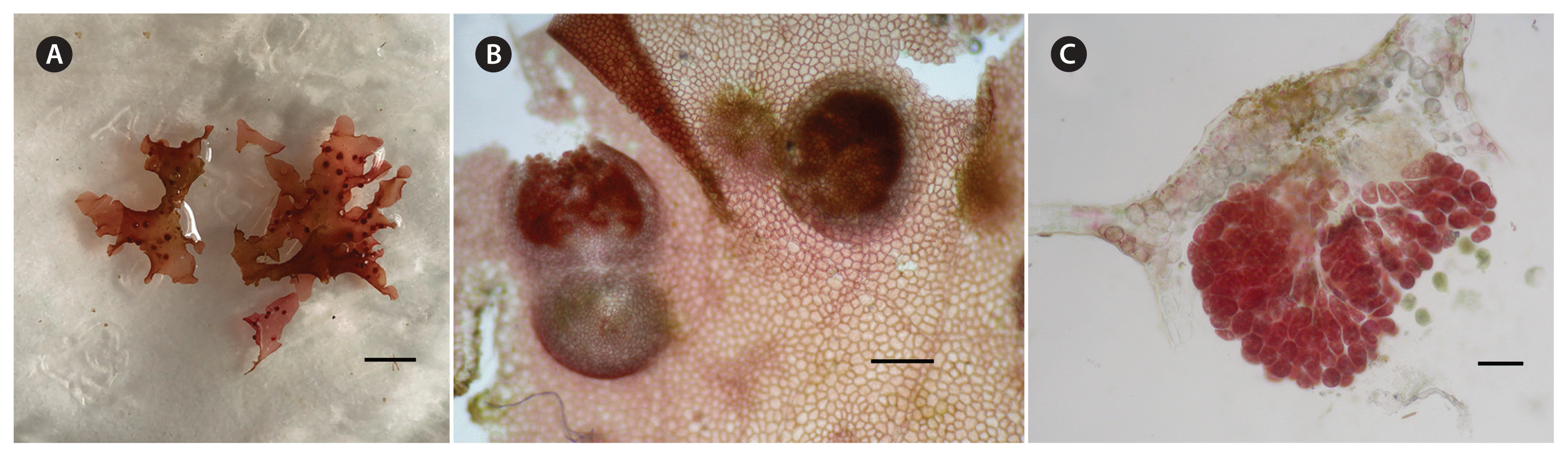
Radicilingua mediterranea sp. nov., female thalli. (A) Habit of female blade with numerous cystocarps. (B) Surface view with cystocarps at different levels of maturity. (C) Transverse section of a mature cystocarp. Scale bars represent: A, 5 mm; B, 200 μm; C, 100 μm.
Diagnosis
Plants 2–4 cm high, pink to red in colour, composed of membranous single-layered blades. Blades erect at the apices, composed of axes with rounded or pointed margins. Branching usually irregular, pseudodichotomously to alternately. Blades traversed by microscopic veins, 1–2 parallel to the central vein. At the apices veins connected by other transverse veins, forming an irregular rectangular pattern. Margin of blades with a large apical cell broad and flat to domelike and narrow in form. Blades anchored to the substratum by a discoid holdfast. Tetrasporangia in irregular sori, scattered over the upper half fertile blade. Cystocarps spherical with a non-protruding ostiole and a two-layered pericarp.
Holotype
Voucher A000840 at PAD Herbarium, Botanical Garden Padova (Italy).
Type locality
Venice Lagoon, Venice, North Adriatic, Italy.
Etymology
The specific epithet (fem. adj.) refers to the area (i.e., the Mediterranean Sea) where it was found up to now.
Molecular voucher
LR991382 (rbcL-5P).
Vegetative morphology
Thalli were 2–4cm high, pink to red in colour (Fig. 3A), often forming aggregates of 10–15 cm, composed of overlapping membranous single-layered blades, 40–50 μm in thickness, tangled and often adherent to each other through marginal unicellular rhizoids (haptera) grouped in bundles that can reach 300–500 μm in length. Blades became erect at the apices, which were composed of axes with rounded or pointed margins. Branching was usually irregular, pseudodichotomously to alternately, resulting in deeply lobed thalli (Fig. 3A). Blades were traversed by microscopic veins, usually 3 cells thick in cross-section, which, in surface view, were composed by a single, rarely double, row of elongate cells, 15–90 μm long × 10–30 μm wide (Fig. 3B), connected by conspicuous pit connections. Veins were mostly parallel, 1–2 lateral veins parallel to a central vein. At the apices of the blade lateral veins were often connected to the central one by other transverse veins, forming an irregular rectangular pattern (Fig. 3B). At the margin of blades, a large apical cell was present (10 μm in diameter) (Fig. 3C), varying in form from broad and flat to domelike and narrow. This initial gave rise to a primary cell row composed of cells arranged in semi-circular arrays around the apical cell (Fig. 3C). Inner surface cells were irregular, rectangular to polygonal, 30–100 μm long × 20–60 μm wide, with conspicuous secondary pit connections, and were characterized by numerous parietal discoid plastids (Fig. 3D). At the margin of the blade the cells were smaller with elongate-elliptical projections (Fig. 3E). Blades were anchored directly to the substratum by a discoid holdfast and secondarily attached by uniseriate, multinucleate rhizoidal filaments extending from marginal surface cells (Fig. 3F & G). New young blades arose from the surface cells of the holdfasts (Fig. 3H).
Reproductive morphology
Tetrasporangia were borne in sori formed over the upper half of the entire thallus on both sides of the blade. Tetrasporangial sori were irregular in shape, scattered over the upper half fertile blade and arranged on both sides of the apical veins (Fig. 4A & B). Mature tetrasporangia were spherical-ellipsoid, tetrahedrally divided, 40–50 μm in diameter (Fig. 4C). Cystocarps were scattered on both sides of the thallus (Fig. 5A). When mature they were spherical, 335–575 μm in diameter with a non-protruding ostiole and a two-layered pericarp (Fig 5B). Carposporangia were ovoid to spherical, 24–39 × 36–78 μm (Fig. 5C).
DISCUSSION
In this study we report the first molecular data for the genus Radicilingua Papenfuss and analyse the phylogenetic relationship between this genus and the morphologically overlapping Calonitophyllum Aregood.
The taxonomic treatment of the two genera has undergone several changes in the past. Holmes (1873) described the type species of the genus Radicilingua as Nitophyllum thysanorhizans Holmes, after analysing northeastern Atlantic Nitophylleae Willkomm from Cornwall, England. In 1924, Kylin moved the species to the genus Rhizoglossum (as Rhizoglossum thysanorhizans [Holmes] Kylin), but Kylin’s Rhizoglossum is an illegitimate name because later homonym of Presl’s (1845) Rhizoglossum. Therefore, later Papenfuss (1956) erected the new genus Radicilingua nomen novum to include the northeastern Atlantic Radicilingua thysanorhizans (Holmes) Papenfuss and two Mediterranean species: Radicilingua adriatica (Kylin) Papenfuss and Radicilingua reptans (Kylin) Papenfuss.
During the same period, on the other side of the Atlantic Ocean (western Atlantic), Hoyt (1920) described Nitophyllum medium from material collected on offshore reefs and in the drift along the coast of North Carolina (USA). Subsequently, Taylor (1960) proposed the combination Hymenena media (Hoyt) W. R. Taylor, based on the superficial resemblance of this veined plant to representatives of the genus Hymenena Greville from South Africa, Pacific North America, and Australia. But, at around the same time, Wagner (1954) reported that the morphology and development of the procarp and carposporophyte of plants in North Carolina revealed no relationship to Hymenena (typified by H. venosa [L.] C. Krauss). Indeed, procarp development of the North Carolina plants was similar to that of Nitophyllum bonnemaisonii (C. Agardh) Greville, a European species, while early apical growth of the blades and subsequent vein formation were similar to that in Radicilingua (= Rhizoglossum) thysanorhizans (Holmes) Papenfuss (Holmes 1873, Kylin 1924, Papenfuss 1956, Huvè and Riouall 1970). To solve this taxonomic issue, Aregood (1975) erected a new monotypic genus for the North Carolina populations: Calonitophyllum Aregood. She reported that C. medium resembled R. thysanorhizans in vegetative developmental pattern, but was distinguishable by very prominent macroscopic veins and by a procarp characterized by a single carpogonial filament and a single large persistent sterile cell. These characters are not easy to observe, especially in immature and vegetative thalli (Aregood 1975). Moreover, macroscopic veins are not present in males and in young thalli and the number of cell layers is not specified (Aregood 1975). On the contrary, Hoyt (1920) described the veins of N. medium as one cell wide, rarely two cells wide. Thus, further morphological analyses of American C. medium are needed to clarify the vein size.
In the light of all this, currently the taxonomic status of these taxa is controversial and distinguishing the two genera is very difficult with the only morphological observations.
In the present study, the rbcL-5P phylogenetic reconstruction shows that the Radicilingua and Calonitophyllum specimens group together with strong statistical support and the rbcL-5P sequence percent identities calculated among the members of this clade are comparable to intrageneric rbcL-5P values for other genera of the subfamily Nitophylloideae (Supplementary Table S2). This suggests that the genera Radicilingua and Calonitophyllum are congeneric, and the name Radicilingua Papenfuss (Papenfuss 1956) has priority. Therefore, we here propose to transfer the only species described so far for the genus Calonitophyllum, C. medium (Aregood 1975), to the genus Radicilingua as Radicilingua media (Hoyt) Wolf, Sciuto and Sfriso comb. nov.
The phylogenetic reconstruction shows that the Mediterranean Radicilingua specimens are genetically distinct from the northeastern Atlantic R. thysanorhizans sampled from the United Kingdom. The rbcL-5P sequence divergences found between the Mediterranean and northeastern Atlantic Radicilingua lineages are comparable to the rbcL-5P interspecific divergences calculated for other genera of the subfamily Nitophylloideae (Supplementary Table S2).
Despite their phylogenetic divergence, our Adriatic and Ionian samples are morphologically very similar to R. thysanorhizans, even if they present the following two differences: (1) a smaller number of lateral parallel veins (1–2 on both sides of the principal vein) in our samples, compared to the myriad of veins forming a trellis-like pattern in R. thysanorhizans; (2) the form of the cystocarp ostiole: non-protruding in our samples, protruding in R. thysanorhizans. Our samples differ also from the other two species of the genus. In fact, R. adriatica, originally described as Rhizoglossum adriaticum by Kylin (1924), can be distinguished from our samples by the smaller size of the thallus (1 cm in R. adriatica, 2–4 cm in the Adriatic and Ionian specimens) and by the presence of a single parallel vein on both sides of the central vein. Moreover, Kylin (1924) did not observe a rectangular pattern of veins at the apices of the blades as present in our samples. R. reptans, in contrast to the other species, is easily recognizable because parallel veins are completely absent (Kylin 1924). It is a very rare species (Sfriso and Curiel 2007) that was reported in the Venice lagoon for the first time only in the 1990s (Curiel et al. 1996).
For these reasons, we attribute the Mediterranean samples to the new species R. mediterranea Wolf, Sciuto & Sfriso. Our results show that R. mediterranea and R. thysanorhizans are not easily distinguishable by a morphological point of view, since they can exhibit overlapping morphological characters, but they are clearly distinct at the species level based on molecular analyses (Fig. 2).
During our sampling surveys, we did not find any specimens of the other Radicilingua species reported for the Mediterranean Sea. Concerning records of R. thysanorhizans, it is possible that, due to the overlapping morphology with our specimens, this Atlantic species has never been present in the Mediterranean Sea and that the previously collected specimens attributed to this taxon were misidentified. Similar misidentifications are not new for red algal specimens collected in the Mediterranean Sea, as reported by previous studies. For example, Wolf et al. (2011) demonstrated that Adriatic samples of Ceramium Roth with characters falling inside the morphological range of one or more Atlantic species of this genus, and previously identified as those species, were indeed phylogenetically distinct from their Atlantic counterparts (Wolf et al. 2011). Recently, based on molecular analyses and an important diacritical morphological character, Serio et al. (2020) recognized and described the new species Melanothamnus testudinis Serio et al., generally overlooked in the Mediterranean records and previously confused with the morphologically very similar Pacific species Polysiphonia carettia Hollenberg.
Concluding, with this study we provide the first molecular data for R. thysanorhizans and we describe the new species R. mediterranea. Moreover, our molecular results demonstrate that the morphologically overlapping genera Radicilingua and Calonitophyllum are congeneric, Radicilingua having priority; so the type species of Calonitophyllum is here transferred to Radicilingua.
SUPPLEMENTARY MATERIALS
Supplementary Table S1. List of the organisms belonging to the family Delesseriaceae whose sequences are included in the phylogenetic analyses based on the rbcL gene (https://www.e-algae.org).
Supplementary Table S2. Percent identity matrix for the 5P-rbcL gene fragment (630 aligned positions) of the subfamily Nitophylloideae (https://www.e-algae.org).
ACKNOWLEDGEMENTS
Funded by Provveditorato Interregionale Opere Pubbliche per il Veneto, Trentino Alto Adige e Friuli Venezia Giulia, through Consorzio Venezia Nuova and CORILA (Consortium for coordination of research activities concerning the Venice lagoon system)—Project “VENEZIA 2021.” We acknowledge Giuseppe Portacci for his contribution to sampling and the anonymous reviewers for their useful suggestions.
Notes
The authors declare that they have no potential conflicts of interest.