Genetic discontinuity of Digenea (Rhodomelaceae, Rhodophyta) from Mexico supports recognition of two new species, D. mexicana and D. rafaelii
Article information
Abstract
Genetic continuity of Digenea simplex outside the Atlantic waters remains less studied, despite its long exposure to oriental folk medicine and wide distribution in tropical waters. Mitochondrial COI-5P and plastid rbcL sequences and morphology were investigated for plants from Mexico and additional specimens from Japan. Two new species, Digenea mexicana and Digenea rafaelii, are described for plants that would previously have been recognized as D. simplex in Mexico. D. mexicana grows to 10.5 cm tall and has cylindrical axes, irregular or di-, trichotomous branches, simple to branched determinate branchlets with eight pericentral cells, and tetrasporangia on inflated upper parts of determinate branchlets. It occurs intertidally in Quintana Roo, Yucatan Peninsula. D. rafaelii grows to 5 cm tall and has cylindrical axes, irregular or di-, trichotomous branches, and simple determinate branchlets with ten pericentral cells. It occurs intertidally in the Gulf of California. Both COI-5P and rbcL sequences revealed the genetic discontinuity between D. mexicana and D. rafaelii.
INTRODUCTION
Digenea C. Agardh is a rhodomelacean red algal genus that until recently was regarded as monospecific, including only Digenea simplex (Wulfen) C. Agardh (Agardh 1822). D. simplex is widely known as one of the most efficient anthelmintic treatments in oriental folk medicine (Tseng 1983, Trono 1997, Ebadi 2006). The species is also of interest as a source of polysaccharides inhibiting inflammatory and nociceptive responses and the production of carrageenan (Takano et al. 2003, Pereira et al. 2014). Since its first recognition in the Adriatic Sea, D. simplex has been recorded from numerous locations and is now regarded as having a pantropical distribution in the northern and southern hemisphere (Pakker et al. 1996, Silva et al. 1996, Orfanidis and Breeman 1999, Guiry and Guiry 2018).
Agardh (1822) established Digenea to accommodate a species previously known as Conferva simplex Wulfen. The genus is characterized by terete, polysiphonous axes, with branches that are densely surrounded by short, stiff, simple filamentous branchlets (Agardh 1822, Kylin 1956, Norris 2014). A second species, Digenea subarticulata Simons, was described for plants from KwaZulu-Natal, South Africa (Simons 1970), however, De Clerck et al. (2005) noted that the species likely belongs to the genus Bryocladia F. Schmitz. Recently, Digenea arenahauriens C. W. Schneider, Hamzeh & G. W. Saunders from Bermuda and D. cymatophila (R. E. Norris) Díaz-Tapia & Maggs from Hawaii have been added in the genus (Díaz-Tapia et al. 2017, Schneider et al. 2018).
The apparent pantropical distribution of D. simplex has been the focus of biogeographical and taxonomic studies. For example, Pakker et al. (1996) found that Pacific isolates of D. simplex survive a broader range of temperatures than Atlantic populations, which may be correlated to local temperature extremes. Orfanidis and Breeman (1999) reported that ecological variation in D. simplex was related to the lower tolerance limits of temperature, indicating that the pantropical distribution may be related to evolutionary pressures. Based on DNA sequences and morphology, Schneider et al. (2018) described D. arenahauriens for plants from Bermuda that were previously known as D. simplex. However, there have been no studies on plants outside the Atlantic, where D. simplex is reported to commonly occur (e.g., Yoshida 1998, Norris 2014).
Plants referable to D. simplex occur abundantly in the intertidal to subtidal zones of the Yucatan Peninsula and the Gulf of California, Mexico (Dawson 1963, Dreckmann and Sentíes 1994, Norris 2014). Quintana Roo on the east of the Yucatan Peninsula is an adjacent rocky coast to the Caribbean Sea, comprising mostly sandy bottom, subtidal rocky platforms exposed to the swell, and rocky-sand substrata in reef lagoons. Compared to many studies on the Gulf of California (e.g., Norris 2010, 2014), little is known about marine algae from Quintana Roo because many coastal areas remain unexplored or no descriptions have been made using recent molecular tools. We focused on the genetic discontinuity between D. simplex specimens from the Yucatan Peninsula and the Gulf of California. We studied morphology and analyzed mitochondrial COI-5P and plastid rbcL sequences, as commonly used in taxonomic studies of red algae (e.g., Boo et al. 2016a, 2016b, Schneider et al. 2018). On the basis of morphology and two genes sequences, we propose two new species of Digenea, one from Yucatan Peninsula and the other from the Gulf of California.
MATERIALS AND METHODS
Samples of Digenea were collected from low intertidal zones of the Yucatan Peninsula and the Gulf of California, Mexico, and additional samples from Japan were included (Supplementary Table S1). Vouchers were mounted on herbarium sheets and subsamples were dehydrated in silica gel for molecular analysis. Herbarium specimens in the Herbario Nacional de México (MEXU) of Universidad Nacional Autónoma de México were examined on loan. Vegetative and reproductive structures were observed under a microscope. For anatomical observations, plants were sectioned using razor blades and were stained with 1% aqueous aniline blue. Photographs were taken with a DP-71 camera (Olympus, Tokyo, Japan) mounted on a BX-51 microscope (Olympus). Types and voucher specimens are housed at the Herbarium of the Department of Biology, Chungnam National University, Daejeon, Korea (CNUK) and MEXU of the Universidad Nacional Autónoma de México. Abbreviations of herbaria follow the online Index Herbariorum (Thiers 2018).
DNA extraction, polymerase chain reaction amplification, and sequencing were performed as described in Boo et al. (2013). The primers used for amplifying and sequencing were rbcLJNF1-rbcLJNR1 and R753-RrbcS start for rbcL (Freshwater and Rueness 1994, Kang and Kim 2013), and GWSFn-GazRx for COI-5P (Saunders and Moore 2013). Sequences of COI-5P and rbcL generated in this study were deposited in GenBank (Benson et al. 2018). A total of 17 COI-5P sequences (664 bp) were aligned, including sequences of other species of Digenea previously published in GenBank. Seventeen rbcL sequences (1,220 bp) were aligned including sequences of other species of Digenea previously published in GenBank and three outgroup taxa, Alsidium corallinum C. Agardh, Bryothamnion seafortii (Turner) Kützing, and B. triquetrum (S. G. Gmelin) M. Howe (Supplementary Table S1). However, we used phylogeny of COI-5P without outgroups because branch lengths of ingroups were too short to recognize each species.
Phylogenies of COI-5P and rbcL datasets were inferred using maximum likelihood (ML) and Bayesian inference (BI). The ML analyses were performed using the Pthreads version of RAxML v8.0.X (Stamatakis 2014) set as follows: a rapid bootstrap analysis and search for the best-scoring ML tree in one single program run with 1,000 bootstrap replicates under GTRGAMMA model. BI was performed for individual datasets with MrBayes v.3.2.1 (Ronquist et al. 2012) using the Metropolis-coupled Markov Chain Monte Carlo (MC3) with the GTR + G + I model. For each matrix, four million generations of two independent runs were performed with four chains and sampling trees every 100 generations. The burn-in period was identified graphically by tracking the likelihoods at each generation to determine whether they reached a plateau. Twenty-five percent of saved trees were removed, and the remaining trees were used to infer Bayesian posterior probabilities (BPP).
RESULTS
Digenea mexicana G. H. Boo & D. Robledo sp. nov. (Fig. 1A–J)

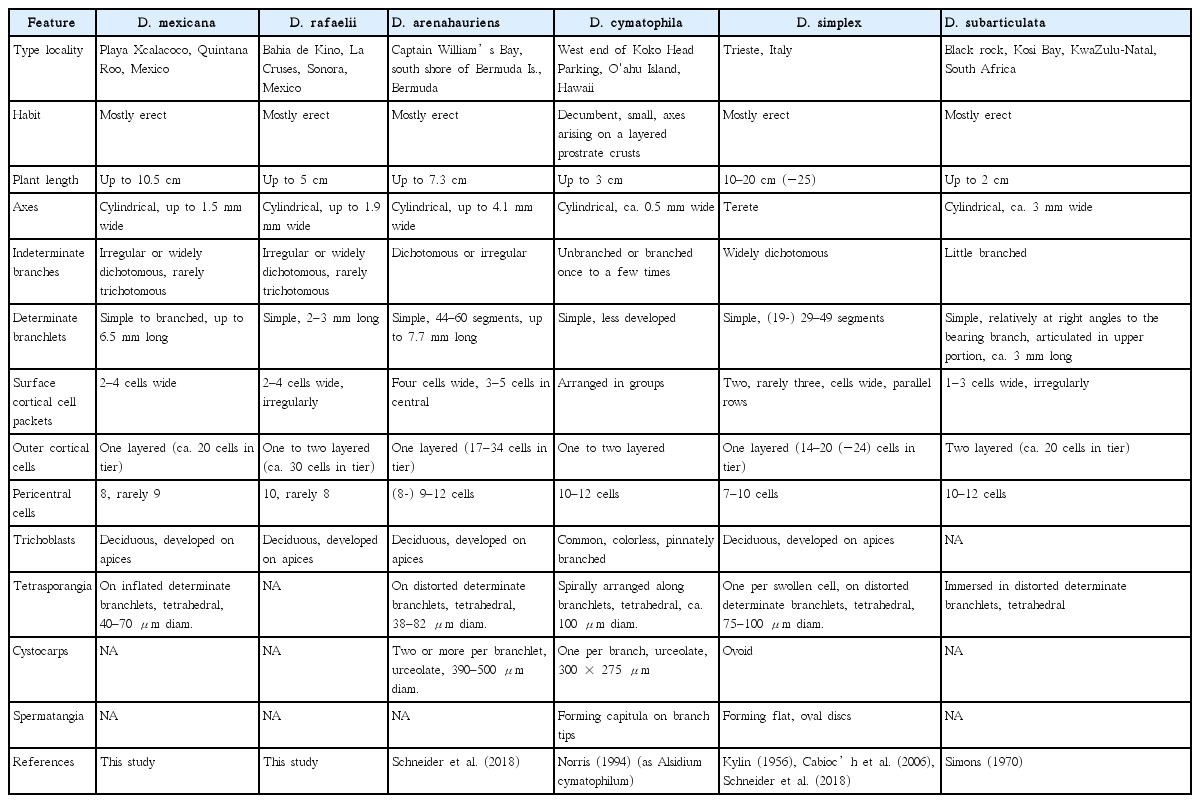
Digenea mexicana sp. nov. G. H. Boo & D. Robledo. (A) Plants growing intertidally on sand covered rocks at Playa Xcalacoco, Quintana Roo, Mexico (type locality). (B) Type specimen (CNU080663). (C) Paratype specimen from Veracruz (MEXU 2326). (D) Alternate determinate branches (CNU080683). (E) Surface view of determinate branch showing cortical cell packets covering pericentral cells (CNU080681). (F) Transverse section of indeterminate branch showing radiating determinate branchlets (CNU080682). (G) Transverse section of determinate branchlet having eight pericentral cells (pc) around axial cell (ax) and a single layer of outer cortical cells (cc) (CNU080682). (H) Transverse section of determinate branchlet having nine pericentral cells (pc) around axial cell (ax) and a single layer of outer cortical cells (cc) (CNU080681). (I) Swollen determinate branchlet with tetrasporangia (arrowheads) (CNU080684). (J) Tetrahedrally divided tetrasporangia (arrowhead) (CNU080684). Scale bars represent: B & C, 2 cm; D, F & I, 100 μm; E, G, H & J, 20 μm.
Description
Plants bushy, up to 10.5 cm high, tough and cartilaginous (Fig. 1A–C); axes sometimes denuded in lower portions, attached by a discoid holdfast which often spreads and may possibly coalesce with other holdfasts; axes terete, irregularly, widely dichotomously or rarely trichotomously branched (Fig. 1B & C); axes and indeterminate branches usually densely (or occasionally sparsely) clothed with wiry, stiff, simple or branched, determinate branchlets of 6.5 mm in length and 90–135 μm in width (Fig. 1F); branchlets polysiphonous, with axial cells 20–30 μm in diameter (Fig. 1D), 8 or rarely 9 pericentral cells surrounded by about 20 cortical cells in one layer (Fig. 1G & H); surface cortical cells oblong, irregularly or longitudinally arranged, in packets of 2–4 cells (Fig. 1E); apices with short vegetative trichoblasts remaining during development; tetrasporangia formed on swollen determinate branchlets, tetrahedrally divided, ellipsoidal to globose, 40–70 μm in diameter (Fig. 1I & J); sexual reproductive structures not found.
Holotype
CNU080663 deposited in CNUK, Herbarium of Chungnam National University, Daejeon, Korea; a tetrasporophyte specimen; collected at Playa Xcalacoco (20°39′42.84″ N, 87°01′56.78″ W), Quintana Roo, Mexico on Apr 19, 2018 by Daniel Robledo.
Isotypes
CNU080664, CNU080665, CNU080666, and CNU080668 in CNUK, Korea.
Additional herbarium specimens observed
MEXU 2310 (as D. simplex determined by José Luis Godínez Ortega; Morro de la Mancha, Veracruz; Nov 8, 2013), MEXU 2326 (as D. simplex determined by José Luis Godínez Ortega; Morro de la Mancha; May 18, 2013), MEXU 2357 (as D. simplex determined by José Luis Godínez Ortega; Morro de la Mancha; Oct 20, 2011), MEXU 2371 (as D. simplex determined by José Luis Godínez Ortega; Mandinga, Veracruz; Apr 7, year unknown), CNU080681 (Playa Xcalacoco, Quintana Roo, Mexico; Jun 16, 2018), CNU080682 (Playa Xcalacoco; Jun 16, 2018), CNU080683 (Playa Xcalacoco; Jun 16, 2018), CNU080684 (Playa Xcalacoco; Jun 16, 2018).
Etymology
The specific epithet refers to Mexico, where the plants were collected.
Distribution and habitat
Presently confirmed in Playa Xcalacoco, Quintana Roo, Yucatan Peninsula and likely extended to the Gulf of Mexico. D. mexicana populations were found growing on intertidal rock, calcareous corallines and reef formations, which are often covered by sands. They formed patches or mats of 1–2 m2 in area (Fig. 1A).
DNA sequences of type specimens
For holotype, MH514874 (COI-5P) and MH514862 (rbcL); for isotypes, MH514875-MH514878 (COI-5P) and MH514863-MH514866 (rbcL).
Digenea rafaelii G. H. Boo, G. Andrade-Sorcia & S. M. Boo sp. nov. (Fig. 2A–F)
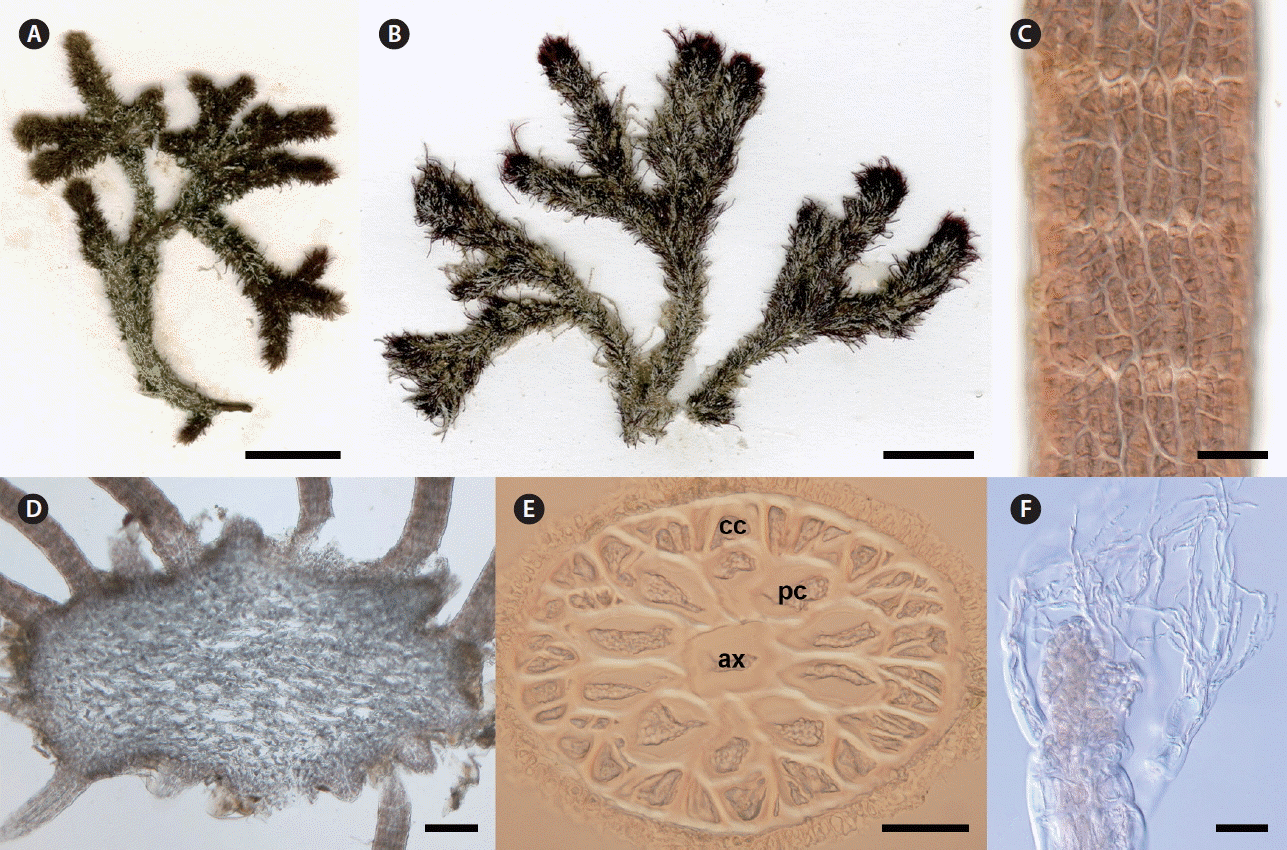
Digenea rafaelii sp. nov. G. H. Boo, G. Andrade-Sorcia & S. M. Boo. (A) Type specimen (CNU037775). (B) Paratype specimen from Conquista Agraria (CNU58249). (C) Surface view of determinate branchlet showing cortical cell packets covering pericentral cells (CNU037775). (D) Transverse section of indeterminate branch showing radiating determinate branchlets (CNU037776). (E) Transverse section of determinate branchlet having ten pericentral cells (pc) around axial cell (ax) and a single layer of outer cortical cells (cc) (CNU037775). (F) Apices with short vegetative trichoblasts (CNU037775). Scale bars represent: A & B, 1 cm; C, E & F, 50 μm; D, 200 μm.
Description
Plants bushy, up to 5 cm high, tough and cartilaginous; axes widely di-, trichotomous or irregularly laterally branched (Fig. 2A & B); axes denuded at base or sparsely covered with wiry branchlets, 1.9 mm wide; branches usually densely (or occasionally sparsely) clothed with slender, stiff, usually simple determinate branchlets, 2–3 mm long and 98–270 μm wide (Fig. 2D); primary axes sometimes denuded in lower portions; attached by a discoid holdfast which often spreads and may possibly coalesce with other holdfasts; branchlets polysiphonous, with axial cells 30–60 μm in diameter, 10 or rarely 8 pericentral cells surrounded by up to 30 cortical cells in one layer (Fig. 2E); surface cortical cells irregular, in packets of 2–4 cells (Fig. 2C); apices with short vegetative trichoblasts remaining during development (Fig. 2F); reproductive structures not found.
Holotype
CNU037775 deposited in CNUK; a vegetative specimen; collected at Bahia de Kino (28°88′71″ N, 112°04′96″ W), Las Cruces, Sonora, Mexico on May 16, 2010 by Sung Min Boo.
Isotypes
CNU037775-1, CNU037775-2, and CNU037775-3 in CNUK, Korea.
Additional herbarium specimens observed
CNU 058249 (Conquista Agraria, Baja California Sur, Mexico; Feb 27, 2014), CNU058249-2 (Conquista Agraria; Feb 27, 2014), CNU058291 (Cabo San Lucas, Baja California Sur, Mexico; Mar 1, 2014).
Etymology
The specific epithet honors the departed saint Rafael Riosmena-Rodriguez, previous Professor of Departamento de Biología Marina, Universidad Autónoma de Baja California Sur, La Paz, Mexico, for his outstanding contributions to the marine algae in Mexico.
Distribution and habitat
Presently confirmed in Bahia de Kino, Sonora and Cabo San Lucas, Conquista Agraria, and Las Conchas, Baja California Sur. Plants were collected on sand covered rocks in mid to low intertidal or sometimes in sublittoral zones.
DNA sequences of type specimens
For holotype, MH514879 (COI-5P) and MH514867 (rbcL); for isotype, MH514880 (COI-5P) and MH514868 (rbcL).
A morphological comparison of D. mexicana and D. rafaelii with all other species of Digenea is provided in Table 1.
Mitochondrial COI-5P and plastid rbcL phylogenies
Twenty-four sequences were generated in this study: five of COI-5P and rbcL from each new species (here assigned to D. mexicana sp. nov. and D. rafaelii sp. nov.), respectively, and two each of COI-5P and rbcL for Digenea plants from Japan (Supplementary Table S1). All five plants of D. mexicana were identical in COI-5P and rbcL, and plants of D. rafaelii from four locations in the Gulf of California were also the same in both markers. D. mexicana differed by 3.6% in COI-5P and 2.5–2.7% in rbcL from D. rafaelii and by 3.0–3.4% in COI-5P and 0.98–1.07% in rbcL from D. arenahauriens. Digenea plants from Japan and Western Australia differed by 3.9–4.2% in COI-5P from D. simplex from Tunisia.
The topology of the ML and BI trees was largely congruent, and only the ML tree is shown for COI-5P and rbcL (Figs 3 & 4). In the COI-5P phylogeny (Fig. 3), D. mexicana and D. rafaelii were distinct enough to be segregated from other species of Digenea. However, Digenea plants from Japan and Australia did not form a clade with D. simplex from the Mediterranean.

Maximum likelihood (ML) tree inferred from mitochondrial COI-5P sequences of the genus Digenea. ML bootstrap values (≥50%) and Bayesian posterior probabilities (≥0.90) are shown at branches. Dashes indicate values <50 or <0.90. Bold letters indicate newly generated sequences in this study.
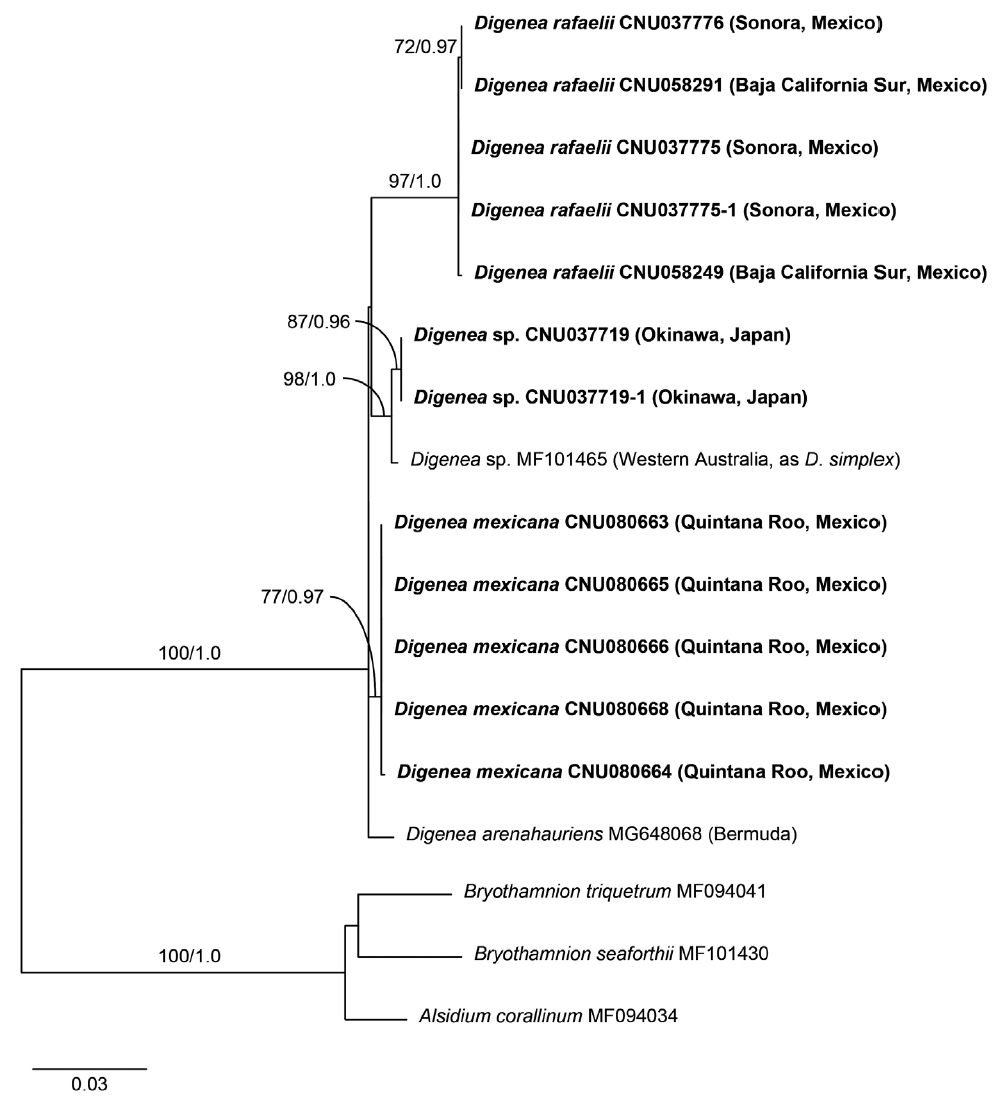
Maximum likelihood (ML) tree inferred from plastid rbcL sequences of the genus Digenea. ML bootstrap values (≥50%) and Bayesian posterior probabilities (≥0.90) are shown at branches. Dashes indicate values <50 or <0.90. Bold letters indicate newly generated sequences in this study.
In the rbcL phylogeny (Fig. 4), the monophyly of the genus Digenea was strongly supported (100% ML, 1.0 BPP). D. rafaelii from the Gulf of California was placed outside the group of D. arenahauriens, D. mexicana, and D. simplex. Phylogenetic relationships of D. mexicana with other species were not resolved.
DISCUSSION
Our results indicate that, based on COI-5P and rbcL sequences, the D. simplex complex comprises more than six genetically distinct species, including D. mexicana sp. nov. and D. rafaelii sp. nov. from Mexico. Interestingly, D. mexicana on the Caribbean side is geographically isolated from D. rafaelii on the Pacific side, as the other species are endemic to their distributional range. All species can be recognized by comparative morphology (Table 1). The habit and size of plants, the presence or absence of indeterminate branches, the number of pericentral cells and the surrounding outer cortical cells, and branching of determinate branchlets can be used as diagnostic characters in segregating species, as discussed below. However, identification of Digenea species without DNA data and / or information on collection sites could prove difficult.
D. mexicana is distinct from D. rafaelii in morphology as well as COI-5P and rbcL sequences. It occurs at Playa Xcalacoco, Quintana Roo on the Yucatan Peninsula. Plants grow to 10.5 cm tall and have cylindrical axes, irregular or widely dichotomous branches of indeterminate growth, simple to branched branchlets of determinate growth, eight pericentral cells in determinate branchlets and tetrasporangia formed in the upper inflated branchlets. Plants from Veracruz are large (about 14.5 cm tall in herbarium specimen of MEXU 2326), as comparable in size to the Mexican plant illustrated by Dreckmann and Sentíes (1994). Tetrasporangia were formed on the distal part of determinate branchlets, as reported in previous studies (Dawson 1963, Dreckmann and Sentíes 1994, Norris 2014). Mexican plants have a monophasic life history including only a tetrasporangial phase (Dreckmann and Sentíes 1994).
D. rafaelii occurs at four locations on the south and north coasts of the Baja California Peninsula. Plants grow to 5 cm tall and have cylindrical axes, irregular or widely dichotomous branches, and simple determinate branchlets with ten pericentral cells. The size of D. rafaelii is much smaller than that of D. mexicana. Plants from Baja California Peninsula have been reported to grow to 25 cm tall (Norris 2014), comparable to that of European plants of D. simplex (Kylin 1956, Cabioc’h et al. 2006), however plants in our collections are usually about 5 cm tall. Our specimens from Puerto Peñasco (collected in May 2010) and La Paz (collected in February 2015) were sterile without tetrasporangia and sexual reproductive structures. Populations of Digenea from the Pacific coasts of Mexico are mostly vegetative in the present and previous collections (Dreckmann and Sentíes 1994), although tetrasporangia were present only in specimens at Bahia de Kino in August and September (Mendoza-González and Mateo-Cid 1986). We could speculate that Baja California coasts is the northern boundary of D. rafaelii, where its populations might be maintained mostly by vegetative reproduction or mitotic tetrasporangia. Range of D. rafaelii might be extended to Islas Revillagigedo and Costa Rican Golfo Dulce, where Digenea plants commonly occur (Taylor 1945, Dawson 1957).
D. arenahauriens (type locality: Captain William’s Bay, Bermuda) grows to 7.3 cm tall and has terete main axes with long wiry determinate branchlets composed of 44–60 axial segments (Schneider et al. 2018). Determinate branchlets grow to 7.7 mm long and have a relatively large axial cell (27–78 μm in diameter) with 9–12 pericentral cells surrounded by 17–34 outer cortical cells. Female plants have two or more cystocarps borne on a determinate branchlet. Tetrasporangia are formed subapically on determinate branchlets, are tetrahedrally divided and 38–82 μm in diameter (Schneider et al. 2018). D. arenahauriens occurs in Bermuda and the Caribbean Antilles (Schneider et al. 2018). Further study of Digenea in the Caribbean Sea will highlight the speciation and biogeographic patterns of D. arenahauriens and D. mexicana.
D. cymatophila (type locality: O’ahu Island, Hawaiian Islands) was transferred from Alsidium based on plastid genome data (Díaz-Tapia et al. 2017). It occurs on rocks in sandy beaches, forming a mass of erect determinate branches from a layered basal crust (Norris 1994). It develops only one cystocarp on each fertile branchlet of female plants. D. cymatophila is the sole decumbent species in Digenea, and the decumbent habit likely arose as a response to grazing pressure by herbivores (Lewis et al. 1987).
D. simplex (type locality: Trieste, Italy; the inner coast of Adriatic Sea) grows to 25 cm tall and has terete axes of indeterminate growth that are irregularly to dichotomously branched (Kylin 1956, Schneider et al. 2018). Determinate branchlets are wiry, with 29–49 segments and a relatively small axial cell (19–38 μm in diameter) and 7–10 pericentral cells surrounded by 14–20 (−24) outer cortical cells (Kylin 1956, Schneider et al. 2018). It occurs throughout the year in the Mediterranean except the Gulf of Lion (Cabioc’h et al. 2006).
D. subarticulata (type locality: Black Rock, Kosi Bay, KwaZulu-Natal, South Africa) is small in size (less than 2 cm tall) and has simple axes with 10–12 pericentral cells (Simons 1970). Determinate branchlets are relatively long and slightly articulated at the upper portion (Simons 1970). De Clerck et al. (2005) noted that D. subarticulata is likely representative of Bryothamnion, because of its small size and occurrence apart from northern KwaZulu-Natal, where is the southern boundary of Digenea.
Both COI-5P and rbcL phylogenies indicate that Digenea plants from Japan and Australia, in the past referred to D. simplex (Yoshida 1998, Huisman and Borowitzka 2003), are different species from D. simplex from Tunisia. Considering that the type locality of D. simplex is Trieste, Mediterranean samples could be considered as typical. The close relationships (1.5% in COI-5P, 0.4% in rbcL) between the Japanese and Australian taxa suggest their recent divergence from a common ancestor, which has likely occurred widely from southern to northern hemispheres. Additional study is needed to unravel the phylogenetic bridge between Western Australia and Japan and to decide whether plants from Australia and Japan belong to the same species or are distinct species.
We dispute the merging of New Caledonian Digenea vieillardii Kützing into D. simplex by De Toni (1903) and Millar and Prud’homme van Reine (2005), who considered the former species as a heterotypic synonym of the latter. Kützing (1865) described D. vieillardii on the basis of specimens that Eugène Vieillard, a French botanist, collected during his expedition to New Caledonia. The diagnosis was “Ramellis ramosis”, and the branched determinate branchlets were well illustrated in Tabulae 28 g, h, I, k, l in the protologue (Kützing 1865). Our present study supports the taxonomic value of branched determinate branchlets in Digenea because it characterizes D. mexicana from D. rafaelii. D. vieillardii has tetrachotomous to verticillate branchlets at the upper part, while determinate branchlets of D. mexicana are mostly formed once. In contrast, D. simplex has simple, unbranched determinate branchlets (Kylin 1956, Schneider et al. 2018). Molecular analysis of type or topotype material of D. vieillardii is necessary to unravel its phylogenetic relationship within Digenea.
This is the first study of the genetic discontinuity between Digenea plants from Mexico, where we describe two new species, D. mexicana in Yucatan Peninsula and D. rafaelii in the Gulf of California. The previous reports of D. simplex in Australia and Japan (Yoshida 1998, Huisman and Borowitzka 2003) are misidentifications due to the cryptic nature of the D. simplex complex. Further global sampling will highlight taxonomy and distribution patterns of the D. simplex complex. Since D. simplex is one of the major medicinal red algae (Ebadi 2006), our finding will contribute to its medicinal exploitation and the conservation of local species.
SUPPLEMENTARY MATERIAL
Information on specimens included in molecular analyses (http://e-algae.org).
ACKNOWLEDGEMENTS
We thank Rafael Riosmena-Rodriguez for his help in collection in Baja California Peninsula, José Luis Godinez-Ortega from MEXU for herbarium on loan, and Marina Morabito for COI-5P sequence of the Tunisian sample. The first author thanks field guiders in the pre-congress tours of 2009 IPC in Okinawa, Japan. This study was supported by a postdoctoral scholarship from the Silva Center for Phycological Documentation, University Herbarium, University of California Berkeley to GHB, PN-Conacyt: 2015-01-575 to DR, and a grant of Marine Biotechnology Program (2017143), Korean Ministry of Oceans and Fisheries to SMB. We appreciate John Huisman and anonymous reviewers for improving our manuscript.