ABSTRACTKelp forests and the many vital ecosystem services they provide are threatened as the severity of climate change and other anthropogenic stressors continues to mount. Particularly in the North Pacific, sea surface temperature is warming and glacial melt is decreasing salinity. This study explored the resiliency of early life-history stages of these foundation species through a factorial laboratory experiment. The effects of rising sea surface temperature under low salinity conditions on kelp spore settlement and initial gametophyte growth in Eualaria fistulosa, Nereocystis luetkeana, and Saccharina latissima were investigated. Decreased settlement and growth were observed in these species at elevated temperatures and at low salinity. Eualaria fistulosa spores and gametophytes were the most negatively impacted, compared to the more widely distributed N. luetkeana and S. latissima. These results suggest that N. luetkeana and S. latissima could potentially outperform E. fistulosa under projected conditions. However, despite decreased performance among all species, our findings indicate that these species are largely resilient to temperature changes when exposed to a low salinity, even when the temperature changes are immediate and extreme. By exploring how early life-history stages of several key kelp species are impacted by dual stressors, this research enhances our understanding of how kelp forests will respond to projected and extreme changes in temperature when already stressed by low salinity.
INTRODUCTIONKelp forests, common features along most temperate rocky coastlines, provide numerous vital ecosystem services. Kelps are foundation species, providing essential nursery and adult habitat for commercially, recreationally and ecologically important fish and invertebrate species (Dayton 1985, Bologna and Steneck 1993, Seitz et al. 2014, Thornton and Herbert 2014). They support a wide range of trophic levels via complex food webs (Schiel and Foster 2006, Lorentsen et al. 2010). Kelp forests also reduce coastal erosion via wave dampening (Neal et al. 2010). Furthermore, they are significantly more productive than many other marine habitats (e.g., open ocean, nearshore sandy habitats) (Duggins et al. 1989). For instance, Macrocystis pyrifera kelp forests in California have an average ecosystem net primary productivity of 2.8 ± 0.6 g C m−2 d−1 (Miller et al. 2011). Additionally, kelp forests play a key role in offshore nutrient cycling and act as a dispersal mechanism for many fish and invertebrate species, with drift kelp providing a vector for larval transport (Hinojosa et al. 2011). Largely because of their important ecosystem functions, it is critical that we assess how kelps may be impacted by changes in ocean condition.
Continued increases in sea surface temperature (SST) negatively impact many marine ecosystems (Hoegh-Guldberg and Bruno 2010). For example, seawater warming trends can result in range shifts of warmer-water species that outcompete resident species, that may already be metabolically stressed under rising temperatures (Ling et al. 2009, Hoegh-Guldberg and Bruno 2010). From 1960 to 1990, global SST has increased by 0.14 ± 0.04°C per decade (Casey and Cornillon 2001). However, it is suspected that this global warming trend is severely underestimated in many nearshore marine systems where increases in SST are projected to be ten times greater, as direct anthropogenic warming of the coastal zone results from nearby urban developments (Amos et al. 2013). On the Gulf of Alaska shelf in particular, records from the last 45 years indicate that the upper 100 m is warming at a rate of ~0.20°C decade−1 (Royer and Grosch 2006, Kelly 2015). If this upward trend continues, an increase of ~2.0°C would be expected in this region in the next century. Additionally, climate change is increasing glacial melt, which lowers salinity of estuarine waters (Dyurgerov and Meier 2005). Modest projections indicate that the North Pacific is expected to see an overall 0.005 decade−1 decrease in salinity (Boyer et al. 2005). On the Gulf of Alaska shelf, salinities in the upper 100 m have decreased by ~0.15 decade−1 since 1970 (Kelly 2015). If this downward trend continues, a ~1.5 decrease in salinity would be expected in this region in the next century. High-latitude coastal regions are already experiencing low salinities, given their proximity to melting glaciers and increases in precipitation, in southern Alaska in particular (Arendt et al. 2009, Bieniek et al. 2014). Furthermore, the northern Gulf of Alaska is home to the largest freshwater discharge system in North America, with a 62% greater freshwater input than that of the Mississippi River (Royer 1982, Atkinson and Grosch 1999). Freshwater discharge in North Pacific coastal systems is not only driving salinity down, but also increasing sediment suspension that will affect photosynthetic capacity and possibly further intensify impacts of rising temperatures and decreasing salinities on kelp forest communities (Wiencke et al. 2007, Spurkland and Iken 2011).
Climate change induced stressors, such as increasing temperatures and decreasing salinities, are likely to play key roles in shaping kelp forests in the near future. Rising seawater temperatures have been linked to kelp forests undergoing phase shifts, resulting in less biodiverse, less productive algal-turf-dominated systems or urchin barrens (Connell and Russell 2010, Moy and Christie 2012, Krumhansl et al. 2014). Metabolic adjustments in some kelp species, while allowing them to persist in the face of warming waters, have resulted in an overall decrease in resilience due to reduced ecological performance by kelp recruits (Wernberg et al. 2010). This is likely because microscopic, young stages of kelp species are more susceptible to temperature and salinity fluctuations than their mature counterparts (Fredersdorf et al. 2009). The negative impacts of temperature and salinity on kelp (e.g., inhibition of photosynthetic activity and reduction of germination capacity) occur to a greater extent under the concurrent stress of both factors, as they are thought to interact synergistically (Fredersdorf et al. 2009). Furthermore, it is important to examine species-specific responses to these dual stressors, as the species of kelp that comprise a forest can greatly impact associated community composition. For example, sites with both canopy and understory kelp species offer greater structural complexity than those with understory species alone, and are capable of supporting higher densities of fish accordingly (Hamilton and Konar 2007). Although generally negative impacts of rising temperatures and decreasing salinities on kelp forests are supported by some studies, the specifics of how many key North Pacific kelp species will perform and fare under these projected conditions remains unknown (Fredersdorf et al. 2009, Connell and Russell 2010, Wernberg et al. 2010, Krumhansl et al. 2014).
Spores represent a critical life-stage for algal species as both dispersal and settlement occurs through the spore stage (Reed 1990). Spores must surpass obstacles (e.g., changes in temperature and salinity, physical barriers, strong currents, and over-shading by mature species) during the critical settlement and recruitment periods. The successful settlement of spores directly contributes to long-term community variability, as their dispersal, settlement, and recruitment aid in determining the future distribution of mature individuals. If rising temperature along with low salinity alters, or even hinders, the ability of spores to settle and grow into gametophytes, the effects will likely be reflected in long-term community composition. By exploring how early life-history stages of several key species are impacted by dual stressors, this research enhances our understanding of how kelps will respond to projected and extreme changes in temperature coincident with low salinity, which may have important repercussions for the diverse ecosystems that these kelps support.
Study speciesThe most common understory kelp species in the study region is Saccharina latissima, and the two predominant canopy kelp species are Nereocystis luetkeana and Eualaria fistulosa. The understory kelp, S. latissima is considered to be perennial, though some populations exhibit blade degradation in the winter, leaving only the stipes until spring when the blades regrow (Lindeberg and Lindstrom 2010). S. latissima distribution ranges from the Arctic Ocean to Santa Catalina Island, California, and it can also be found in Russia, Japan, and Korea (Lindeberg and Lindstrom 2010). Sori form as dark patches on the blades of this understory species.
The two canopy-forming species, N. luetkeana and E. fistulosa, are the only canopy-forming species present at the study site’s high latitude (59° N). The biannual N. luetkeana is the most prevalent canopy kelp throughout most of central and south-central Alaska, with its range extending from the eastern Aleutians (Umnak Island) to San Luis Obispo, California (Lindeberg and Lindstrom 2010). Sori form on surface blades of N. luetkeana and, after reaching maturity, drop to the benthos where spores are released. E. fistulosa is another common biannual kelp in central Alaska and is the only canopy-forming kelp in much of the Aleutian Islands. Its distribution ranges from Russia/Japan to southern Southeast Alaska (Lindeberg and Lindstrom 2010). Sori develop on the reproductive sporophylls of E. fistulosa, which can be found on the upper stipe near the seafloor.
MATERIALS AND METHODSStudy site descriptionKachemak Bay, Alaska is an elongated embayment contiguous with the southeastern entrance to Cook Inlet. The bay is 63 km long and 39 km wide at its entrance. It experiences an 8.7 m tidal range and input from seven glaciers along its southern shore (Field and Walker 2003). Regional circulation is characterized by an inflow of generally cyclonic ocean currents from the Gulf of Alaska. These currents enter Kachemak Bay primarily along its southern shore. Freshwater runoff from the surrounding glaciers and watersheds dilute the salinity and increase the sediment load as water circulates within the bay, before exiting along the north shore (Field and Walker 2003). Between 2002 and 2014, SST in Kachemak Bay had a mean summer high (based on the average temperature during the single warmest 24-hour period, June–August of each year) of 12.2°C and a total annual range of −1.9°C to 13.6°C (water quality data) (NOAA National Estuarine Research Reserve System 2002–2014). Between 2002 and 2014, salinity in Kachemak Bay had a mean summer low (based on the average salinity during the single least saline 24-h period, June–August of each year) of 27.9 and a total annual range of 23.6 to 34.0 (water quality data) (NOAA National Estuarine Research Reserve System 2002–2014).
Factorial spore experimentTo determine the effects of temperature and salinity on kelp spore settlement success and compare physiological thresholds between common North Pacific kelp species (N. luetkeana, E. fistulosa, and S. latissima), a laboratory experiment was conducted during the summer of 2015. For the purposes of this study, settlement success was defined in terms of spore settlement density (at 24 h, post-spore-release), gametophyte growth (germ tube length at 7 d, post-spore-release), and the proportion of gametophytes to inviable attached spores (at 7 d, post-spore release). Our specific hypotheses were as follows: 1) spore settlement success across study species declines with increasing temperature and decreasing salinity and 2) spore settlement success in E. fistulosa is the most negatively impacted under increasing temperature and decreasing salinity, followed by the more widely distributed species: N. luetkeana, and S. latissima.
Sori (i.e., kelp reproductive structures) were collected during biweekly boat and SCUBA searches for fertile individuals of N. luetkeana, E. fistulosa, and S. latissima. These sori are readily identified as dark patches on the blades of kelp (found at the surface in N. luetkeana, and near the benthos in E. fistulosa and S. latissima). Blades with ripe sori were removed from target kelp individuals and placed in a seawater-filled container for transport back to the laboratory. Since sori of these species do not all ripen concurrently (e.g., N. luetkeana typically ripens earlier in the summer than E. fistulosa), experimental trials were conducted throughout the summer (Jun–Aug).
In the laboratory, collected sori were lightly scrubbed and rinsed with 0.9-μm filtered seawater to remove impurities, such as macroscopic epiphytes or excess mucilage. Spores were then released in synchrony before each trial, following the methods described by Deiman et al. (2012). For this, the sori were packed in a cooler between layers of damp paper towels and plastic wrap and kept in the dark at 9°C for 30–60 min. Sori were then removed from the cooler and rinsed with 0.9-μm filtered seawater to remove any prematurely released spores, before being placed in individual beakers filled with 1.0 L of filtered seawater. Sori then remained at 9°C in these beakers under fluorescent light (250 lux) for 30–60 min. The solutions in the beakers became clouded with released spores and mucilage during this period. After using a cheesecloth to remove excess mucilage, the concentrations of resulting spore solutions were quantified using a hemocytometer (with a minimum detectable concentration of 2,000 spores mL−1). All spore solutions were ultimately diluted to final concentrations of 4,000 spores mL−1 in 400-mL volumes (1.6 million spores total, per trial) and placed in individual open-top plastic containers before experimental trials began.
One-half of the experimental solutions were diluted using 0.9-μm filtered seawater and remained at a seasonally ambient salinity of 31. Remaining spore solutions were diluted with a mixture of 0.9-μm filtered seawater and 0.9-μm filtered fresh water, resulting in a salinity of 26 (a typical low and stressful salinity for this region). Salinity was monitored and maintained throughout all experimental trials using a hand-held refractometer (±1 accuracy). The refractometer was calibrated using deionized water prior to use. Following dilutions, three clear glass microscope slides were placed at the bottom of each spore solution container to provide settlement substrate, after which spore solutions (with glass slides) were placed in temperature-controlled water baths under fluorescent light (250 lux). Individual containers were allowed to gradually rise to a given treatment temperature (13, 17, and 21°C, or current, projected, and extreme highs, respectively) over the next 1–2 h (or remain the same in the case of the 9°C/ambient controls).
Spore solutions remained exposed to temperature and salinity treatment conditions (Fig. 1) as spores were allowed to settle onto the glass slides for 24 h, consisting of an 18-h period of light exposure (the average summer day length in Kachemak Bay) and 6 h of dark. The glass slides (three per trial) were then rinsed with 0.9-μm filtered seawater to remove any unattached spores. Settlement success was quantified under a compound microscope. Quantification was accomplished by haphazardly selecting five fields of view per slide at 400× magnification (0.17 mm2) and counting the number of attached spores and/or gametophytes.
Settlement slides were then returned to the same experimental conditions that they had previously been subjected to, this time in filtered seawater or seawater-freshwater solutions (no longer spore solutions). Gametophytes remained under experimental conditions for an additional 6 d, after which their short-term growth (at 7 d post-spore-release) was quantified. Initial growth was measured from the outside of the original spore cell to the tip of the germ tube on each individual gametophyte. ImageJ software (National Institutes of Health, Bethesda, MD, USA) was used to process images of five haphazardly selected fields of view per slide at 400× magnification (0.17 mm2). As an additional measure of performance, the proportion of gametophytes to inviable attached spores (i.e., no germ tube present) was also recorded 7 d after spore release (Cie and Edwards 2008). In total, five experimental settlement runs were conducted, per kelp species, under all eight combinations of temperature and salinity treatments (n = 5).
Statistical analysesTo determine the relative spore settlement success of each kelp species under the various temperature and salinity treatments, spore settlement concentrations and germ tube lengths were first averaged for each species under each of the eight treatments and standard errors were determined. Percentages of gametophytes versus inviable attached spores present at one week were also averaged between trials for each species at each given treatment.
To test hypothesis one, that spore settlement success across study species declines with increasing temperature and decreasing salinity, two separate two-way ANOVAs were performed. These ANOVAs compared spore settlement concentrations and germ tube growth, with experimental treatments and kelp species as the sources of variation in both cases. Three additional one-way ANOVAs were employed to test the effects of temperature and salinity on each of the three kelp species, separately. Response variables examined here were also spore settlement concentration and germ tube length.
To test which species’ settlement success is the most negatively impacted under increased temperature and decreased salinity scenarios (hypothesis two), t-tests on observations of spore settlement concentrations and germ tube length were conducted. Three t-tests in total were performed, in reference to all possible species combinations (i.e., E. fistulosa vs. N. luetkeana, E. fistulosa vs. S. latissima, and N. luetkeana vs. S. latissima). These comparisons were made for all eight experimental combinations of temperature and salinity conditions. p-values were considered significant at the α = 0.017 level, based on a post-hoc Bonferroni correction.
RESULTSEffects of temperature and salinity on kelp spore settlement success
Eualaria fistulosa spore settlement exhibited a significant decrease in concentrations, from ~15 spores per field of view under the ambient treatment (9°C/31 salinity) to ~5 spores and fewer under more extreme treatments (13°C and greater, both salinities) (Fig. 2A). More gradual, but still significant, declines in spore settlement under increased temperature and decreased salinity were observed in N. luetkeana and S. latissima (Fig. 2B & C). Nereocystis luetkeana spore settlement was highest under the ambient treatment (9°C/31 salinity) with ~24 spores per field of view, and never dropped below ~5 spores per field of view (Fig. 2B). Saccharina latissima spore settlement was also highest under the ambient treatment (9°C/31 salinity) with ~17 spores per field of view, and only fell below ~5 spores under the most extreme treatment (12°C/26 salinity) (Fig. 2C).
Average germ tube length at 7 d in E. fistulosa declined from 20.95 μm under the ambient treatment (9°C/31 salinity) to less than 5 μm under more extreme treatments (17 and 21°C, both salinities) (Fig. 3A). By contrast, N. luetkeana and S. latissima gametophytes exhibited germ tubes longer than 5 μm under all treatment conditions except the most extreme (12°C/26 salinity), at which average germ tube length was 4.95 μm and 2.66 μm, respectively (Fig. 3B & C). Settlement concentration and germ tube length significantly declined across all three species as temperature increased and salinity decreased (from the ambient treatment of 9°C/31 salinity, to the extreme treatments of 17 and 21°C, both salinities) (Figs 2 & 3). However, N. luetkeana gametophyte growth peaked at ~23 μm under the 13°C treatments, before declining significantly (Fig. 3B).
Experimental treatment and kelp species both played statistically significant roles in shaping spore settlement success, defined as spore settlement concentration (p < 0.001 for both factors) and gametophyte growth/germ tube length (p < 0.001 and p = 0.020 for treatment and species, respectively) (Table 1). The interactions of experimental treatments and kelp species were an insignificant factor in terms of shaping spore settlement concentrations and germ tube length, with p-values of 0.283 and 0.253, respectively (Table 1). The effect of variations in temperature was significant for spore settlement concentration amongst all three study species (p < 0.001 for both E. fistulosa and S. latissima, and p < 0.001 for N. luetkeana) (Table 2). The effect of variations in temperature was also found to be significant for germ tube growth in all three study species (p < 0.001 for all three species) (Table 2). Additionally, variation in salinity had a significant effect on spore settlement concentration in S. latissima (p < 0.001) and on germ tube length across all three species (p = 0.003, p = 0.025, and p < 0.001 for E. fistulosa, N. luetkeana, and S. latissima, respectively) (Table 2).
As a further assessment of settlement success, proportions of gametophytes to attached inviable spores were significantly greater in N. luetkeana and S. latissima compared to E. fistulosa, under the 13°C/26 salinity treatment and both 17°C treatments (Fig. 4). However, under the most extreme treatments (21°C/26 salinity and 21°C/31 salinity), there were no significant differences between N. luetkeana and E. fistulosa. Eualaria fistulosa was the only species to produce no gametophytes under experimental conditions (21°C/31 salinity).
Nereocystis luetkeana displayed significantly higher spore settlement densities compared to E. fistulosa under both salinity conditions at increased temperatures of 13, 17, and 21°C (Table 3, Fig. 2A & B). Nereocystis luetkeana similarly displayed significantly greater germ tube lengths compared to E. fistulosa under 13°C/26 salinity, and 17 and 21°C at both salinities (Table 3, Fig. 3A & B). Likewise, S. latissima displayed significantly higher spore settlement densities compared to E. fistulosa under both salinity treatments at 13 and 17°C (Table 3, Fig. 2A & C). Both species performed poorly under 21°C treatments. Saccharina latissima also exhibited significantly greater germ tube lengths under all experimental conditions, except the most extreme treatment of 21°C/26 salinity (Table 3, Fig. 3A & C). Both species performed equally poorly under 21°C/26 salinity.
When compared to each other, N. luetkeana and S. latissima displayed no significant differences in spore settlement and gametophyte growth under nearly all treatments. However, N. luetkeana spore settlement concentration was significantly higher under 21°C/31 salinity (Table 3, Fig. 2B & C). Saccharina latissima germ tube length, on the other hand was significantly greater than N. luetkeana germ tube length under conditions of 9°C/26 salinity (Table 3, Fig. 3B & C).
DISCUSSIONThis study suggests that under projected and extreme increases in temperature coincident with low salinity, the responses of North Pacific kelps are species-specific. Early life-history stages of species with narrower ranges (distribution restricted to higher latitudes) appear to be more vulnerable to ocean warming. Although spore settlement and gametophyte growth generally decreased across the study species as conditions became increasingly stressful, even in the face of extreme and immediate change these species showed great resilience. For example, under low salinity (26) and extreme temperature (21°C) all species still exhibited some degree of settlement and initial growth, which suggests possible resilience to climate change.
Dispersal and settlement of kelps occur through the critical early life-history stages (e.g., spores and gametophytes) (Reed 1990). Accordingly, successful recruitment is key in shaping the distribution and abundance of adult populations. Any effects, positive or negative, of temperature and salinity regimes on early life-history stages will directly impact resulting adult populations, ultimately shaping long-term community assemblages. In a controlled laboratory setting, the early life-history stages of E. fistulosa, N. luetkeana, and S. latissima experienced significant negative impacts under projected and extreme changes in temperature under low salinity conditions. Both spore settlement concentration, proportion of gametophytes to inviable attached spores, and germ tube growth decreased across E. fistulosa, N. luetkeana, and S. latissima under increasing temperature coincident with a low salinity. The degree of decline in settlement success is species-specific, with N. luetkeana and S. latissima generally outperforming E. fistulosa. Other studies have shown similarly negative impacts of climate change related stressors on kelp early life-history stages. For instance, increased levels of pCO2 and temperature have been linked to increased kelp spore mortality and decreased germination rates in a related species, Macrocystis pyrifera (Gaitán-Espitia et al. 2014). Other species of macroalgae, however, experienced strong negative impacts of sedimentation on recruitment success but no temperature-induced effects on recruitment, survival, or growth (Irving et al. 2009). Such varied findings highlight the necessity to explore the species-specific nature of interactions between macroalgae and environmental stressors.
Outperformance of E. fistulosa by N. luetkeana and S. latissima in terms of spore settlement concentration, proportion of gametophytes to inviable attached spores, and gametophyte growth, under increased temperatures at a low salinity could be attributable to E. fistulosa’s relatively narrow range. The population of E. fistulosa examined in this study was near the southern extent of its range (southern Southeast Alaska), as opposed to the more widely distributed N. luetkeana and S. latissima (found as far south as Central and Southern California, respectively). This imbalance in distribution could be a result of inherent differences in the ability of these species to cope with changes in latitudinal gradients of temperature and salinity. Differences in the latitudinal ranges of adult macroalgae have been linked to variations in thermal tolerance, as species with lower latitude distributions were found to be more heat-tolerant while species with a higher latitude terminus to their range were more cold-tolerant (Smith and Berry 1986). The relatively far-reaching southern range of N. luetkeana could also help to explain why gametophyte growth peaked under the moderately warmed treatment of 13°C, as this would be a more typical environmental condition at its lower, mid-range latitudes. Why spore settlement in N. luetkeana does not also follow this trend remains unclear. Gametophyte growth in S. latissima may not follow the same trend since, though it shares a similar southern range limit to that of N. luetkeana, its range extends further north, into the Arctic.
Vulnerability to environmental obstacles/stressors is often life-stage specific (Fredersdorf et al. 2009). Therefore, although spores and gametophytes show great resilience to immediate change, additional work concerning impacts of environmental change on mature life-stages in a natural setting is essential to understanding how resilient these species truly are. Tracking annual species biomass in a natural setting reflects the ability of these populations to gradually adapt to environmental change. Significant quantitative differences in physiological responses to environmental stressors have been observed in geographically isolated populations of kelp, and it has been suggested that climatic change may select for ecotypes that are best suited to changing environmental regimes (Kopczak et al. 1991).
In addition to examining impacts on multiple life-history stages, effects of stressors beyond temperature and salinity should also be explored to form a more complete understanding of resilience in these kelps. Other site-specific factors such as sedimentation, light limitation, wave exposure, and biotic interactions (e.g., herbivory) may play additional roles in shaping these populations. For example, light limitation due to chronic fog cover has been suggested as a key factor in determining the abundance and distribution of Nereocystis in the Aleutians, as opposed to thermal constraints alone (Miller and Estes 1989). Another study implicated herbivory as a factor that can overshadow the role of an abiotic driver, in this case wave action, in determining the distribution and abundance of kelps (Duggins et al. 2001). Variability associated with different types of habitats has also been shown to influence kelp spore production and fecundity (Edwards and Konar 2012).
Since kelps are foundation species, they provide complex structure necessary to support diverse associated communities of a wide array of both ecologically and commercially important taxa (Dayton 1985, Bologna and Steneck 1993, Seitz et al. 2014, Thornton and Herbert 2014). Environmental impacts on key taxa, such as foundation species, are known to result in cascading effects on the community at large due to ecological interactions (Schiel et al. 2004). Therefore, impacts to kelp populations are likely to have indirect effects, altering kelp forest ecosystems as a whole. For instance, future change that results in declines of canopy kelps could have a positive indirect impact on understory species due to competitive release, since canopy species are known to typically inhibit the growth of understory species through domination of light and space (Irving and Connell 2006, Russell 2007). Additionally, differences in fish communities can be linked to presence and type of marine vegetation available in a given habitat (Hamilton and Konar 2007). Different kelp assemblages provide varied levels of protection from predators and attract different prey, which in turn affects the diversity of fishes supported by the ecosystem (Dean et al. 2000). Hence, insights into how ecosystem engineers, like kelps, will be impacted by projected environmental change are invaluable in assessing how the distribution and abundance of associated species are likely to transform through time. This improved understanding of which species will be most impacted under multiple climate change induced stressors can aid in the design of effective management strategies by targeting the preservation of more vulnerable species and the foundation species they rely on to persist.
ACKNOWLEDGEMENTSWe would like to thank Dr. Matt Edwards and Dr. Seth Danielson for their invaluable input throughout the course of this study. This research was supported by the University of Alaska Fairbanks, Gulf Watch Alaska, and the Robert and Kathleen Byrd award.
Fig. 1A schematic of the factorial design spore experiment, illustrating the experimental treatment combinations that zoospores of the three study species were subjected to (n = 5). Ambient summer seawater temperature and salinity of the study site are indicated. 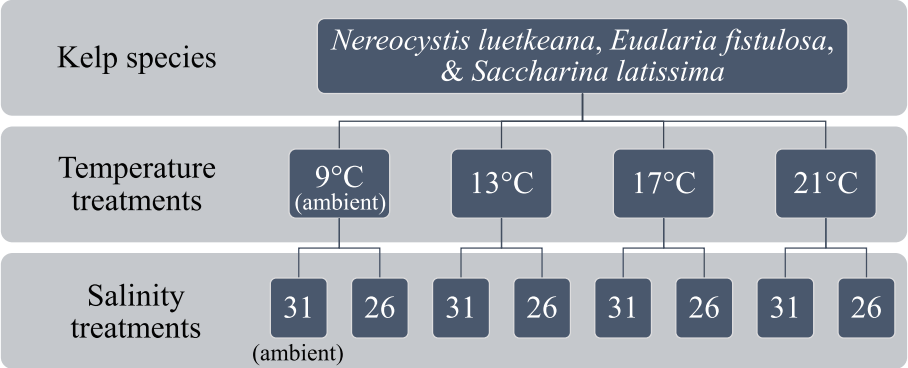
Fig. 2Effects of temperature (9, 13, 17, and 21°C) and salinity (26 and 31) treatments on spore settlement concentration are shown for Eualaria fistulosa (A), Nereocystis luetkeana (B), and Saccharina latissima (C) at 24-h post-spore-release. Concentrations represent the mean ± standard error quantity of attached spores per field of view at 400× (field of view area = 0.17 mm2) for each species under each combination of experimental conditions (n = 5). 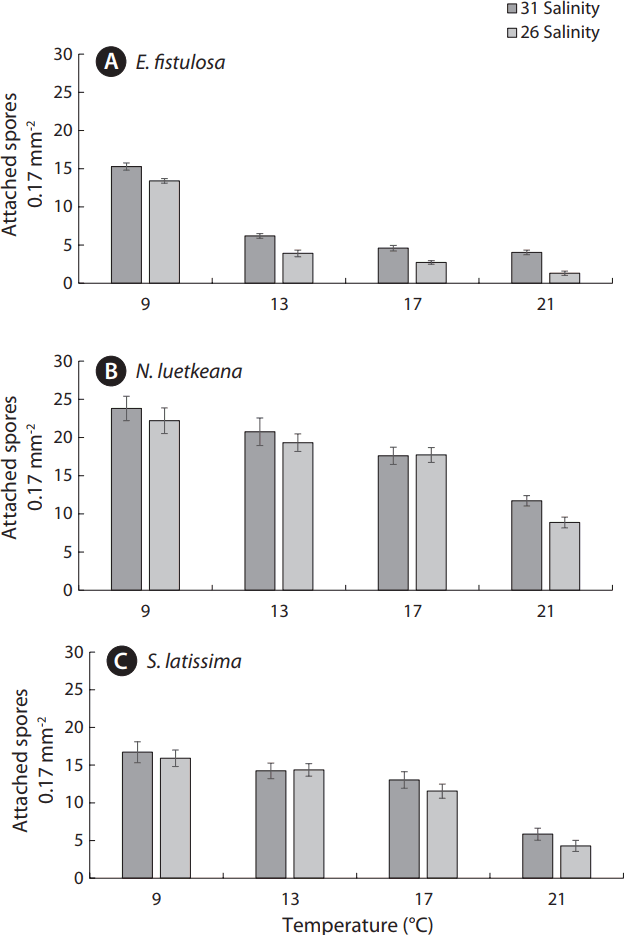
Fig. 3Effects of temperature (9, 13, 17, and 21°C) and salinity (26 and 31) treatments on germ tube length are shown for Eualaria fistulosa (A), Nereocystis luetkeana (B), and Saccharina latissima (C) at 7 d, post-spore-release. Values represent the mean ± standard error germ tube length measured for each species under each combination of experimental conditions (n = 5). 
Fig. 4Effects of temperature (9, 13, 17, and 21°C) and salinity (26 and 31) treatments on proportions of gametophytes, versus nonviable attached spores, are shown for Eualaria fistulosa (A), Nereocystis luetkeana (B), and Saccharina latissima (C) at 24 h, post-spore-release. Values represent the average percent of gametophytes vs. spores for each species under each combination of experimental conditions (n = 5). Letters indicate intraspecific and interspecific significant differences, with progression in alphabetical order corresponding to decreased performance (i.e., greater percent of inviable individuals). 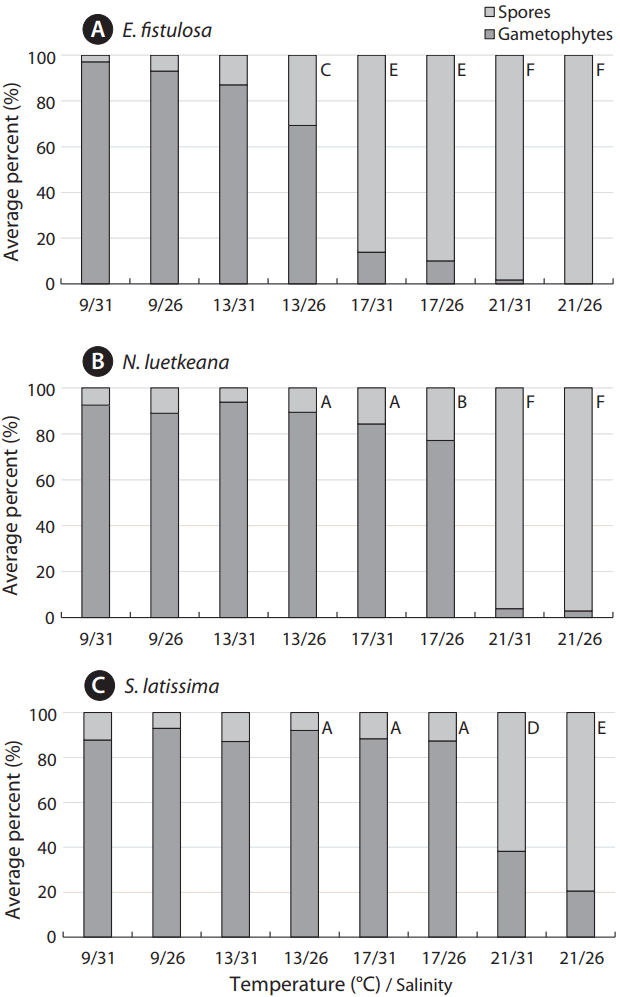
Table 1Two-way analyses of variance (ANOVA) results Table 2Results of ANOVAs on effects of temperature (9, 13, 17, and 21°C) and salinity (26 and 31) on Eualaria fistulosa (EF), Nereocystis luetkeana (NL), and Saccharina latissima (SL) Table 3Results of t-test on paired observations of spore settlement concentrations and germ tube length of Eualaria fistulosa (EF) vs. Nereocystis luetkeana (NL), EF vs. Saccharina latissima (SL) and NL vs. SL REFERERNCESAmos, CL., Al-Rashidi, TB., Rakha, K., El-Gamily, H. & Nicholls, RJ. 2013. Sea surface temperature trends in the coastal ocean. Curr Dev Oceanogr. 6:1–13.
Arendt, A., Walsh, J. & Harrison, W. 2009. Changes of glaciers and climate in northwestern North America during the late twentieth century. J Clim. 22:4117–4134.
Atkinson, LP. & Grosch, CE. 1999. Continental runoff and effects on the North Atlantic Ocean subtropical mode water. Geophys Res Lett. 26:2977–2980.
Bieniek, PA., Walsh, JE., Thoman, RL. & Bhatt, US. 2014. Using climate divisions to analyze variations and trends in Alaska temperature and precipitation. J Clim. 27:2800–2818.
Bologna, PAX. & Steneck, RS. 1993. Kelp beds as habitat for American lobster Homarus americanus. Mar Ecol Prog Ser. 100:127–134.
Boyer, TP., Levitus, S., Antonov, JI., Locarnini, RA. & Garcia, HE. 2005. Linear trends in salinity for the World Ocean, 1955–1998. Geophys Res Lett. 32:L01604 pp.
Casey, KS. & Cornillon, P. 2001. Global and regional sea surface temperature trends. J Clim. 14:3801–3818.
Cie, DK. & Edwards, MS. 2008. The effects of high irradiance on the settlement competency and viability of kelp zoospores. J Phycol. 44:495–500.
Connell, SD. & Russell, DR. 2010. The direct effects of increasing CO2 and temperature on non-calcifying organisms: increasing the potential for phase shifts in kelp forests. Proc R Soc B. 277:1409–1415.
Dean, TA., Haldorson, L., Laur, DR., Jewett, SC. & Blanchard, A. 2000. The distribution of nearshore fishes in kelp and eelgrass communities in Prince William Sound, Alaska: associations with vegetation and physical habitat characteristics. Environ Biol Fish. 57:271–287.
Deiman, M., Iken, K. & Konar, B. 2012. Susceptibility of Nereocystis luetkeana (Laminariales, Ochrophyta) and Eualaria fistulosa (Laminariales, Ochrophyta) spores to sedimentation. Algae. 27:115–123.
Duggins, D., Eckman, JE., Siddon, CE. & Klinger, T. 2001. Interactive roles of mesograzers and current flow in survival of kelps. Mar Ecol Prog Ser. 223:143–155.
Duggins, DO., Simenstad, CA. & Estes, JA. 1989. Magnification of secondary production by kelp detritus in coastal marine ecosystems. Science. 245:170–173.
Dyurgerov, MB. & Meier, MF. 2005. Glaciers and the changing earth system: a 2004 snapshot. INSTAAR Occasional Paper. 58:1–117.
Edwards, MS. & Konar, B. 2012. A comparison of Dragon Kelp, Eualaria fistulosa, (Phaeophyceae) fecundity in urchin barrens and nearby kelp beds throughout the Aleutian Archipelago. J Phycol. 48:897–901.
Field, C. & Walker, C. 2003. A site profile of the Kachemak Bay Research Reserve: a unit of the National Estuarine Research Reserve System. Kachemak Bay Research Reserve, Homer, AK, 134 pp.
Fredersdorf, J., Müller, R., Becker, S., Wiencke, C. & Bischof, K. 2009. Interactive effects of radiation, temperature and salinity on different life history stages of the Arctic kelp Alaria esculenta (Phaeophyceae). Oecologia. 160:483–492.
Gaitán-Espitia, JD., Hancock, JR., Padilla-Gamiño, JL., Rivest, EB., Blanchette, CA., Reed, DC. & Hofmann, GE. 2014. Interactive effects of elevated temperature and pCO2 on early-life-history stages of the giant kelp Macrocystis pyrifera. J Exp Mar Biol Ecol. 457:51–58.
Hamilton, J. & Konar, B. 2007. Implications of substrate complexity and kelp variability for south-central Alaskan nearshore fish communities. Fish Bull. 105:189–196.
Hinojosa, IA., Rivadeneira, MM. & Thiel, M. 2011. Temporal and spatial distribution of floating objects in coastal waters of central–southern Chile and Patagonian fjords. Cont Shelf Res. 31:172–186.
Hoegh-Guldberg, O. & Bruno, JF. 2010. The impact of climate change on the world’s marine ecosystems. Science. 328:1523–1528.
Irving, AD., Balata, D., Colosio, F., Ferrando, GA. & Airoldi, L. 2009. Light, sediment, temperature, and the early life-history of the habitat forming alga Cystoseira barbata. Mar Biol. 156:1223–1231.
Irving, AD. & Connell, SD. 2006. Physical disturbance by kelp abrades erect algae from the understorey. Mar Ecol Prog Ser. 324:127–137.
Kelly, JB. 2015. An examination of hydrography and sea level variability in the Gulf of Alaska. MS thesis. University of Alaska, Fairbanks, AK, 119 pp.
Kopczak, CD., Zimmerman, RC. & Kremer, JN. 1991. Variation in nitrogen physiology and growth among geographically isolated populations of the giant kelp, Macrocystis pyrifera (Phaeophyta). J Phycol. 27:149–158.
Krumhansl, KA., Lauzon-Guay, JS. & Scheibling, RE. 2014. Modeling effects of climate change and phase shifts on detrital production of a kelp bed. Ecology. 95:763–774.
Lindeberg, MR. & Lindstrom, SC. 2010. Field guide to seaweeds of Alaska. Alaska Sea Grant College Program, University of Alaska Fairbanks, Fairbanks, AK, 185 pp.
Ling, SD., Johnson, CR., Frusher, SD. & Ridgway, KR. 2009. Overfishing reduces resilience of kelp beds to climate-driven catastrophic phase shift. Proc Natl Acad Sci U S A. 106:22341–22345.
Lorentsen, S-H., Sjøtun, K. & Grémillet, D. 2010. Multi-trophic consequences of kelp harvest. Biol Conserv. 143:2054–2062.
Miller, KA. & Estes, JA. 1989. Western range extension for Nereocystis luetkeana in the North Pacific Ocean. Bot Mar. 32:535–538.
Miller, RJ., Reed, DC. & Brezezinski, MA. 2011. Partitioning of primary production among giant kelp (Macrocystis pyrifera), understory macroalgae, and phytoplankton on a temperate reef. Limnol Oceanogr. 56:119–132.
Moy, FE. & Christie, H. 2012. Large-scale shift from sugar kelp (Saccharina latissima) to ephemeral algae along the south and west coast of Norway. Mar Biol Res. 8:309–321.
Neal, EG., Hood, E. & Smikrud, K. 2010. Contribution of glacier runoff to freshwater discharge into the Gulf of Alaska. Geophys Res Lett. 37:L06404 pp.
NOAA National Estuarine Research Reserve System. 2002–2014. System-wide Monitoring Program, Available from: http://www.nerrsdata.org. Accessed May 23, 2014
Reed, DC. 1990. The effects of variable settlement and early competition on patterns of kelp recruitment. Ecology. 71:776–787.
Royer, TC. 1982. Coastal fresh water discharge in the Northeast Pacific. J Geophys Res. 87:2017–2021.
Royer, TC. & Grosch, CE. 2006. Ocean warming and freshening in the northern Gulf of Alaska. Geophys Res Lett. 33:L16605 pp.
Russell, BD. 2007. Effects of canopy-mediated abrasion and water flow on the early colonisation of turf-forming algae. Mar Freshw Res. 58:657–665.
Schiel, DR. & Foster, MS. 2006. The population biology of large brown seaweeds: ecological consequences of multiphase life histories in dynamic coastal environments. Annu Rev Ecol Evol Syst. 37:343–372.
Schiel, DR., Steinbeck, JR. & Foster, MS. 2004. Ten years of induced ocean warming causes comprehensive changes in marine benthic communities. Ecology. 85:1833–1839.
Seitz, RD., Wennhage, H., Bergström, U., Lipcius, RN. & Ysebaert, T. 2014. Ecological value of coastal habitats for commercially and ecologically important species. ICES J Mar Sci. 71:648–665.
Smith, CM. & Berry, JA. 1986. Recovery of photosynthesis after exposure of intertidal algae to osmotic and temperature stresses: comparative studies of species with differing distributional limits. Oecologia. 70:6–12.
Spurkland, T. & Iken, K. 2011. Kelp bed dynamics in estuarine environments in subarctic Alaska. J Coast Res. 27:133–143.
Thornton, TF. & Herbert, J. 2014. Neoliberal and neo-communal herring fisheries in Southeast Alaska: reframing sustainability in marine ecosystems. Mar Policy. 61:366–375.
|
|
|||||||||||||||||||||||||||||||||||||||||